| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
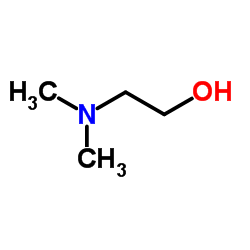 |
2-(二甲基氨基)乙醇
CAS:108-01-0 |
|
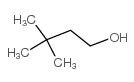 |
3,3-二甲基-1-丁醇
CAS:624-95-3 |
|
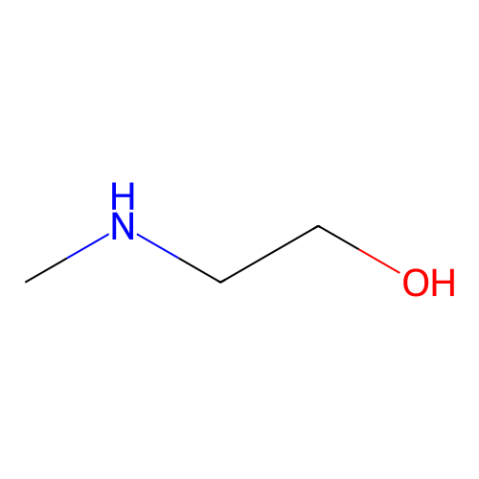 |
2-甲胺乙醇
CAS:109-83-1 |