| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
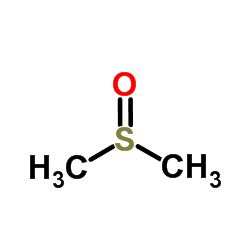 |
二甲基亚砜
CAS:67-68-5 |
|
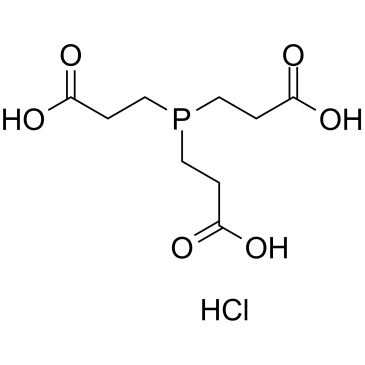 |
三(2-羰基乙基)磷盐酸盐
CAS:51805-45-9 |
|
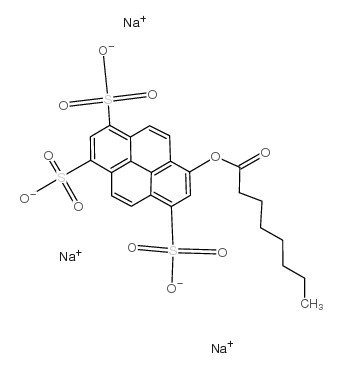 |
8-辛酰氧基芘-1,3,6-三磺酸三钠盐
CAS:115787-84-3 |
|
 |
N,N-二甲基-1-萘胺
CAS:86-56-6 |