Formation and properties of smooth muscle myosin 20-kDa light chain-skeletal muscle myosin hybrids and photocrosslinking from the maleimidylbenzophenone-labeled light chain to the heavy chain.
K N Rajasekharan, J I Morita, M Mayadevi, M Ikebe, M Burke
文献索引:Arch. Biochem. Biophys. 288(2) , 584-90, (1991)
全文:HTML全文
摘要
Experimental conditions which permit the exchange of smooth muscle 20-kDa light chain into skeletal muscle myosin are described. The hybridization does not result in the regulation of actin-activated ATPase activity of the hybrid myosin by smooth light chain phosphorylation. Further, the KCl dependence of the Mg-ATPase activity of the hybrid was similar to that of skeletal muscle myosin. The dephosphorylation of the smooth light chain in the hybrid did not induce a conformational change in the hybrid from the 6 S to the 10 S state, thereby indicating that the conformational transition is dependent also on the nature of the heavy chain subunit. Exchange of the smooth light chain premodified at its Cys-108 by photolabile 4-(N-maleimido)benzophenone and photolysis resulted in crosslinking to the heavy chain subunit. Immunopeptide mapping using a monoclonal antibody against residues 1-23 at the N-terminus of the skeletal muscle myosin heavy chain identified the location of the photocrosslinking site to be beyond 92 kDa away from the N-terminus.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
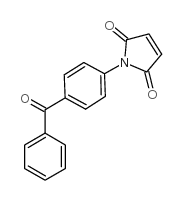 |
4-(N-马来酰亚胺基)二苯甲酮
CAS:92944-71-3 |
C17H11NO3 |
|
Subunit interactions in the clathrin-coated vesicle vacuolar...
1999-10-08 [J. Biol. Chem. 274(41) , 28909-15, (1999)] |
|
Using a low denaturant model to explore the conformational f...
2012-02-21 [Biochemistry 51(7) , 1369-79, (2012)] |
|
A cross-linking study of the N-terminal extension of human c...
2003-09-02 [Biochemistry 42(34) , 10324-32, (2003)] |
|
Identification of the site of photocross-linking formed in t...
1991-02-05 [J. Biol. Chem. 266(4) , 2272-5, (1991)] |
|
The amino-terminal fragment of gelsolin is cross-linked to C...
1992-04-13 [FEBS Lett. 301(1) , 99-102, (1992)] |