| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
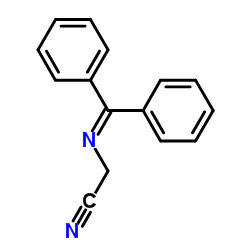 |
二苯亚甲基氨基乙腈
CAS:70591-20-7 |
| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
二苯亚甲基氨基乙腈
CAS:70591-20-7 |