Comparative metabolism of the tobacco-related carcinogens benzo[a]pyrene, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol, and N'- nitrosonornicotine in human hepatic microsomes.
M E Staretz, S E Murphy, C J Patten, M G Nunes, W Koehl, S Amin, L A Koenig, F P Guengerich, S S Hecht
文献索引:Drug Metab. Dispos. 25(2) , 154-62, (1997)
全文:HTML全文
摘要
We compared the metabolism in human hepatic microsomes of three tobacco smoke carcinogens believed to be involved in the induction of cancer in humans: benzo[a]pyrene (BaP),4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK), and N'-nitrosonomicotine (NNN). The metabolism of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL), a major metabolite of NNK, was also investigated. Although the metabolism of some of these compounds by human enzymes or tissue preparations has been previously examined in some studies, they have never been compared in the same human hepatic samples. Moreover, there have been no previous reports of NNAL metabolism by human tissues or enzymes. The tritium-labeled carcinogens (3 microM) were incubated with 10 different human hepatic microsomal preparations and cofactors for 10-20 min, and the products were analyzed by radioflow HPLC. NNN was the best substrate for oxidative metabolism, with the 5'-hydroxylation pathway being the predominant one observed (mean +/- SD = 31 +/- 17 pmol/min/mg protein). alpha-Hydroxylation of NNK by the methylene and methyl hydroxylation metabolic activation pathways was the next fastest reaction, with rates of 3.1 +/- 1.9 and 3.3 +/- 1.1 pmol/min/mg protein, respectively. Metabolism of BaP resulted in the formation of dihydrodiols and phenols; trans-7,8-dihydro-7,8-dihydroxy-BaP, its major proximate carcinogen, was formed at a rate of 1.1 +/- 0.61 pmol/min/mg protein. alpha-Hydroxylation of NNAL proceeded at a rate of 0.53 +/- 0.26 pmol/min/mg protein. The results of this study demonstrate that human hepatic microsomes metabolize all of these tobacco carcinogens resulting in a substantial stream of electrophilic intermediates capable of binding to DNA. The relative rates of oxidative metabolism to electrophiles or their precursors were NNN > NNK > BaP > NNAL. Correlation studies indicated involvement of cytochrome P4502A6 in the 5'-hydroxylation of NNN and cytochrome P4503A4 in the alpha-methylene hydroxylation and pyridine-N-oxidation of NNK and NNAL. The results of this study provide the first data on the comparative metabolism of these important carcinogens in human hepatic microsomes.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
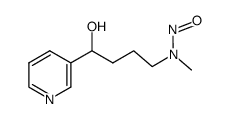 |
4-(甲基亚硝胺)-1-(3-吡啶基)-1-丁醇
CAS:76014-81-8 |
C10H15N3O2 |
|
Quantitation of metabolites of 4-(methylnitrosamino)-1-(3-py...
2002-01-01 [Cancer Res. 62(1) , 129-34, (2002)] |
|
Evidence for endogenous formation of N'-nitrosonornicotine i...
2009-01-01 [Nicotine Tob. Res. 11(1) , 99-105, (2009)] |
|
Preparation of pyridine-N-glucuronides of tobacco-specific n...
2001-05-01 [Chem. Res. Toxicol. 14(5) , 555-61, (2001)] |
|
Effects of long term dietary phenethyl isothiocyanate on the...
1997-09-01 [Carcinogenesis 18(9) , 1715-22, (1997)] |
|
Use of precision-cut tissue slices in organ culture to study...
2000-04-03 [Toxicology 144(1-3) , 83-91, (2000)] |