Bioequivalence of two oral formulations of triflusal capsules in healthy volunteers.
Emilio García Quetglas, Miguel Angel Campanero, Belén Sádaba, Manuel Escolar, Jose Ramón Azanza
文献索引:Arzneimittelforschung 58(6) , 283-7, (2008)
全文:HTML全文
摘要
Triflusal (CAS 322-79-2) is an antiplatelet agent related to salicylates used in several European and Latin American countries in the treatment of cardiovascular diseases. The aim of this paper was to evaluate the bioequivalence of triflusal derived from two preparations using both parent drug and metabolite pharmacokinetic data. The bioavailabolity was measured in 24 healthy male Caucasian volunteers following a single oral dose (600 mg) of the test or reference products in the fasting state. Blood samples were collected for 120 h. Plasma concentrations of triflusal and its metabolite 3-hydroxy-4-trifluoromethylbenzoic acid (HTB) were analyzed by high-performance liquid chromatography with UV and fluorescence detection, respectively. The non-compartmental method was used for pharmacokinetic analysis. Log-transformed Cmax, AUC0-t and AUC0-infinity were tested for bioequivalence using ANOVA and Schuirmann's two-one sided t-test. Tmax was analyzed by nonparametric pharmacokinetic parameters of triflusal and HTB derived from the two formulations were nearly consistent with previous observations. Triflusal parameters derived from the test and reference drug were as follows: Cmax (16.85 +/- 11.41 vs 14.48 +/- 7.22 mg/l), AUC0-t (18.43 +/- 10.91 vs 16.22 +/- 7.58 mg/l per hour), Tmax (1 range 0.25-2h vs 0.875 range 0.25-1.5 h), and t(1/2) (0.49 +/- 00.27 vs 0.76 +/- 0.64). HTB parameters after test and reference formulation administration were as follows: Cmax (68.13 +/- 23.05 vs 65.51 +/- 19.44 mg/l), AUC0-t (2748.18 +/- 971.91 vs 2877.97 +/- 881.2 h x mg/l), AUC0-infinity (3350.15 +/- 1182.62 vs 3372.49 +/- 1110.35 h x mg/l), Tmax (2 range 1-10 h vs 2 range 0.75-12 h), and t(1/2) (42.19 +/- 7.82 vs 43.13 +/- 6.56 h). 90% of confidence intervals for the test/reference ratio of Cmax AUC0-t and AUC0-infinity derived from both triflusal and HTB were found within the range of 80%-125% acceptable for bioequivalence. No significant difference was found between the Tmax values for triflusal and HTB. It was concluded that the two preparations are bioequivalent and may be prescribed interchangeably.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
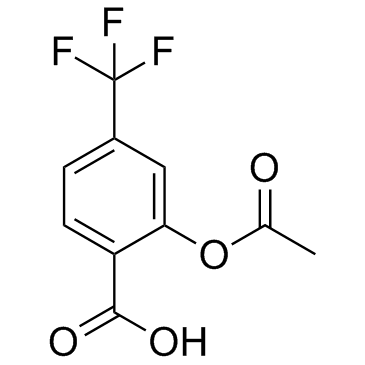 |
三氟醋柳酸
CAS:322-79-2 |
C10H7F3O4 |
|
Long-term follow-up of atrial fibrillation patients in the N...
2009-09-01 [Rev. Esp. Cardiol. 62(9) , 992-1000, (2009)] |
|
Electrochemical behavior of triflusal, aspirin and their met...
2010-08-01 [Comb. Chem. High Throughput Screen 13(7) , 569-77, (2010)] |
|
A phase I study to characterize the multiple-dose pharmacoki...
2011-12-01 [Expert Opin. Drug Metab. Toxicol. 7(12) , 1471-9, (2011)] |
|
Comparative bioavailability study of triflusal oral solution...
2010-01-01 [Arzneimittelforschung 60(1) , 36-41, (2010)] |
|
Gastrointestinal safety of triflusal solution in healthy vol...
2011-07-01 [Eur. J. Clin. Pharmacol. 67(7) , 663-9, (2011)] |