The effects of colchicine analogues on the reaction of tubulin with iodo[14C]acetamide and N,N'-ethylenebis(iodoacetamide).
M C Roach, S Bane, R F Ludueña
文献索引:J. Biol. Chem. 260 , 3015, (1985)
全文:HTML全文
摘要
We have previously found (Ludueña, R. F., and Roach, M. C. (1981b) Biochemistry 20, 4444-4450) that colchicine and podophyllotoxin inhibit the alkylation of tubulin by iodo[14C]acetamide and the formation of an intrachain cross-link in the beta-tubulin subunit by N,N'-ethylenebis(iodoacetamide) (EBI). It was not clear whether these effects were due to conformational changes in tubulin induced by drugs or to direct steric blockage of the sulfhydryl groups involved. In an effort to characterize further these phenomena, we have examined the effects of single-ring and bicyclic analogues of colchicine on the reaction of tubulin with iodo[14C]acetamide and EBI. We have found that neither the A-ring analogues, 3,4,5-trimethoxybenzyl alcohol, 3,4,5-trimethoxybenzaldehyde, 2,3,4-trimethoxybenzaldehyde, and benzaldehyde, nor the C-ring analogues, tropolone and tropolone methyl ether, inhibited alkylation. In contrast, colchicine, podophyllotoxin, and nocodazole and the bicyclic analogues, 5-(2',3',4'-trimethoxyphenyl)-2-methoxytropone and combretastatin, inhibited tubulin alkylation. Since the presence of a bond joining the A and C rings seems to be the determining factor in the suppression of alkylation, it is likely that inhibition by colchicine of the reaction with iodo[14C] acetamide is due largely to a conformational change induced by colchicine. A different pattern was obtained when the effects on cross-link formation by EBI were examined. Here, all the A-ring analogues, the bicyclic analogues, and colchicine, podophyllotoxin, and nocodazole all inhibited formation of the cross-link, whereas the C-ring analogue tropolone methyl ether did not inhibit cross-link formation. Since compounds whose effect on alkylation is markedly different have the same effect on cross-link formation, it is possible that this effect is a steric one and that perhaps the A-ring of colchicine binds to tubulin very close to one of the sulfhydryls involved in the intrachain cross-link formed by EBI in beta-tubulin.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
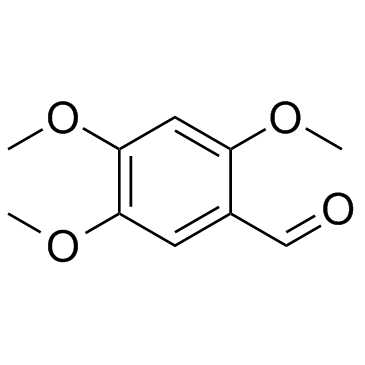 |
2,4,5-三甲氧基苯甲醛
CAS:4460-86-0 |
C10H12O4 |
|
The synthesis of the pyranonaphthoquinones dehydroherbarin a...
2012-10-14 [Org. Biomol. Chem. 10(38) , 7809-19, (2012)] |
|
The effectiveness of Salmonella strains TA100, TA102 and TA1...
1998-01-01 [Mutagenesis 13(1) , 19-26, (1998)] |
|
Enhancement of 4-acetylantroquinonol B production by supplem...
2013-09-25 [J. Agric. Food Chem. 61(38) , 9160-5, (2013)] |
|
2,4,5-TMBA, a natural inhibitor of cyclooxygenase-2, suppres...
2012-07-25 [J. Agric. Food Chem. 60(29) , 7262-9, (2012)] |
|
Compounds from Acorus tatarinowii: determination of absolute...
2010-06-25 [J. Nat. Prod. 73 , 1160-3, (2010)] |