New synthetic substrates of mammalian nucleotide excision repair system.
Alexey Evdokimov, Irina Petruseva, Aleksandra Tsidulko, Ludmila Koroleva, Inna Serpokrylova, Vladimir Silnikov, Olga Lavrik
文献索引:Nucleic Acids Res. 41 , e123, (2013)
全文:HTML全文
摘要
DNA probes for the studies of damaged strand excision during the nucleotide excision repair (NER) have been designed using the novel non-nucleosidic phosphoramidite reagents that contain N-[6-(9-antracenylcarbamoyl)hexanoyl]-3-amino-1,2-propandiol (nAnt) and N-[6-(5(6)-fluoresceinylcarbamoyl)hexanoyl]-3-amino-1,2-propandiol (nFlu) moieties. New lesion-imitating adducts being inserted into DNA show good substrate properties in NER process. Modified extended linear nFlu- and nAntr-DNA are suitable for estimation of specific excision activity catalysed with mammalian whole-cell extracts. The following substrate activity range was revealed for the model 137-bp linear double-stranded DNA: nAnt-DNA ≈ nFlu-DNA > Chol-DNA (Chol-DNA--legitimate NER substrate that contains non-nucleoside fragment bearing cholesterol residue). In vitro assay shows that modified DNA can be a useful tool to study NER activity in whole-cell extracts. The developed approach should be of general use for the incorporation of NER-sensitive distortions into model DNAs. The new synthetic extended linear DNA containing bulky non-nucleoside modifications will be useful for NER mechanism study and for applications.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
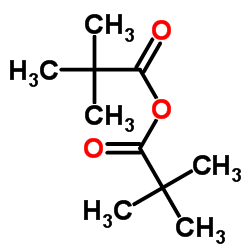 |
特戊酸酐
CAS:1538-75-6 |
C10H18O3 |
|
Kinetic resolution of racemic 2-hydroxy-γ-butyrolactones by ...
2013-03-15 [Org. Lett. 15(6) , 1170-3, (2013)] |
|
The concept of superactive esters. Could peptide synthesis b...
1994-03-01 [Int. J. Pept. Protein Res. 43 , 312, (1994)] |
|
Observation and elimination of N-acetylation of oligonucleot...
2001-05-07 [Bioorg. Med. Chem. Lett. 11(9) , 1105-1107, (2001)] |
|
Structural Basis for Substrate Specificity in Adenosylcobala...
2015-11-06 [J. Biol. Chem. 290 , 26882-98, (2015)] |
|
Kinetic resolution of the racemic 2-hydroxyalkanoates using ...
2010-01-04 [Chemistry 16(1) , 167-72, (2010)] |