Organic & Biomolecular Chemistry
2011-10-07
Control of chemoselectivity in Dieckmann ring closures leading to tetramic acids.
Yong-Chul Jeong, Muhammad Anwar, Tuan Minh Nguyen, Benjamin Song Wei Tan, Christina Li Lin Chai, Mark G Moloney
文献索引:Org. Biomol. Chem. 9 , 6663, (2011)
全文:HTML全文
摘要
An efficient strategy for the control of the chemoselectivity in Dieckmann ring closures leading to tetramic acids derived from serine and α-methyl serine is reported, and this provides pathways to diversely substituted systems from a common starting material.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
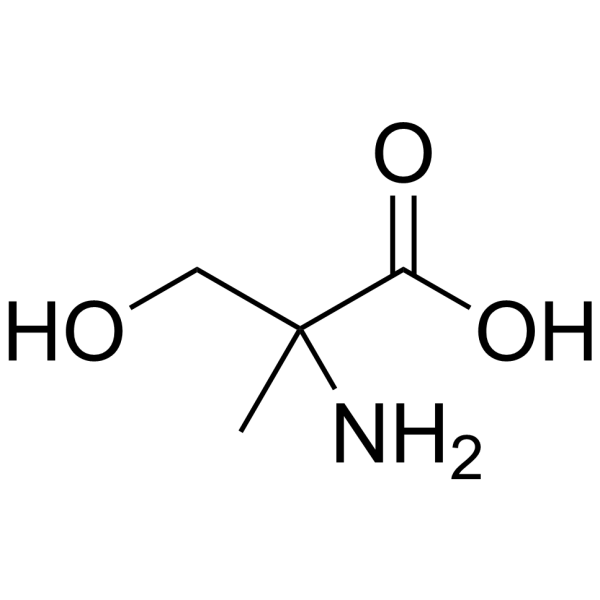 |
2-甲基-DL-丝氨酸
CAS:5424-29-3 |
C4H9NO3 |
相关文献:
更多...
|
Drug design, in vitro pharmacology, and structure-activity r...
2009-08-27 [J. Med. Chem. 52 , 5093-107, (2009)] |
|
The requirement of phosphorylation on a threonine residue in...
1993-09-01 [J. Steroid Biochem. Mol. Biol. 46(3) , 337-47, (1993)] |
|
Synthesis, NMR spectra and function of peptides with alpha-m...
1992-10-01 [Acta Chem. Scand. 46(10) , 989-93, (1992)] |
|
Conformational studies on host-guest peptides containing chi...
1988-11-01 [Int. J. Pept. Protein Res. 32(5) , 344-51, (1988)] |
|
An approach to enantioselective activation of N-benzoyl-alph...
2006-01-01 [Acta Pol. Pharm. 63(5) , 426-9, (2006)] |