Binding of diverse environmental chemicals with human cytochromes P450 2A13, 2A6, and 1B1 and enzyme inhibition.
Tsutomu Shimada, Donghak Kim, Norie Murayama, Katsuhiro Tanaka, Shigeo Takenaka, Leslie D Nagy, Lindsay M Folkman, Maryam K Foroozesh, Masayuki Komori, Hiroshi Yamazaki, F Peter Guengerich
文献索引:Chem. Res. Toxicol. 26(4) , 517-28, (2013)
全文:HTML全文
摘要
A total of 68 chemicals including derivatives of naphthalene, phenanthrene, fluoranthene, pyrene, biphenyl, and flavone were examined for their abilities to interact with human P450s 2A13 and 2A6. Fifty-one of these 68 chemicals induced stronger Type I binding spectra (iron low- to high-spin state shift) with P450 2A13 than those seen with P450 2A6, i.e., the spectral binding intensities (ΔAmax/Ks ratio) determined with these chemicals were always higher for P450 2A13. In addition, benzo[c]phenanthrene, fluoranthene, 2,3-dihydroxy-2,3-dihydrofluoranthene, pyrene, 1-hydroxypyrene, 1-nitropyrene, 1-acetylpyrene, 2-acetylpyrene, 2,5,2',5'-tetrachlorobiphenyl, 7-hydroxyflavone, chrysin, and galangin were found to induce a Type I spectral change only with P450 2A13. Coumarin 7-hydroxylation, catalyzed by P450 2A13, was strongly inhibited by 2'-methoxy-5,7-dihydroxyflavone, 2-ethynylnaphthalene, 2'-methoxyflavone, 2-naphththalene propargyl ether, acenaphthene, acenaphthylene, naphthalene, 1-acetylpyrene, flavanone, chrysin, 3-ethynylphenanthrene, flavone, and 7-hydroxyflavone; these chemicals induced Type I spectral changes with low Ks values. On the basis of the intensities of the spectral changes and inhibition of P450 2A13, we classified the 68 chemicals into eight groups based on the order of affinities for these chemicals and inhibition of P450 2A13. The metabolism of chemicals by P450 2A13 during the assays explained why some of the chemicals that bound well were poor inhibitors of P450 2A13. Finally, we compared the 68 chemicals for their abilities to induce Type I spectral changes of P450 2A13 with the Reverse Type I binding spectra observed with P450 1B1: 45 chemicals interacted with both P450s 2A13 and 1B1, indicating that the two enzymes have some similarty of structural features regarding these chemicals. Molecular docking analyses suggest similarities at the active sites of these P450 enzymes. These results indicate that P450 2A13, as well as Family 1 P450 enzymes, is able to catalyze many detoxication and activation reactions with chemicals of environmental interest.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
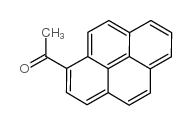 |
1-乙酰基芘
CAS:3264-21-9 |
C18H12O |
|
One-pot synthesis of 1-acetylpyrene over supported phosphotu...
[React. Kinet., Mech. Catal. 108(2) , 531-544, (2013)] |
|
Fundamental photoluminescence properties of pyrene carbonyl ...
[Tetrahedron 68(31) , 6177-6185, (2012)] |
|
1-Acetylpyrene with dual functions as an environment-sensiti...
[Tetrahedron 66(52) , 9798-9807, (2010)] |
|
1-(Bromoacetyl) pyrene, a novel photoinitiator for the copol...
[Coll. Polymer Sci. 285(4) , 397-404, (2007)] |
|
Synthesis and fluorescent properties of 5-(1-pyrenylethynyl)...
[Russian J. Bioorg. Chem. 26(1) , 34-44, (2000)] |