Synthesis and bioactivity of natural and C-3 fluorinated biosynthetic precursors of 28-homobrassinolide.
L R Galagovsky, E G Gros, J A Ramírez
文献索引:Phytochemistry 58(6) , 973-80, (2001)
全文:HTML全文
摘要
In this paper we describe the synthesis of two new fluorinated brassinosteroids: (22R,23R)-22,23-dihydroxy-3alpha-fluorostigmastan-6-one and (22R,23R)-22,23-dihydroxy-3beta-fluorostigmastan-6-one. Their bioactivities were evaluated in the rice lamina inclination test and compared with that of 28-homocastasterone, 28-homotyphasterol and 28-homoteasterone, possible biosynthetic precursors of 28-homobrassinolide. Results confirmed expected similarities between the biosynthesis of 24-ethylbrassinosteroids (named as the 28-homo series) and that described for 24-methylbrassinosteroids, and also indicated that these precursors might exhibit per se activities.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
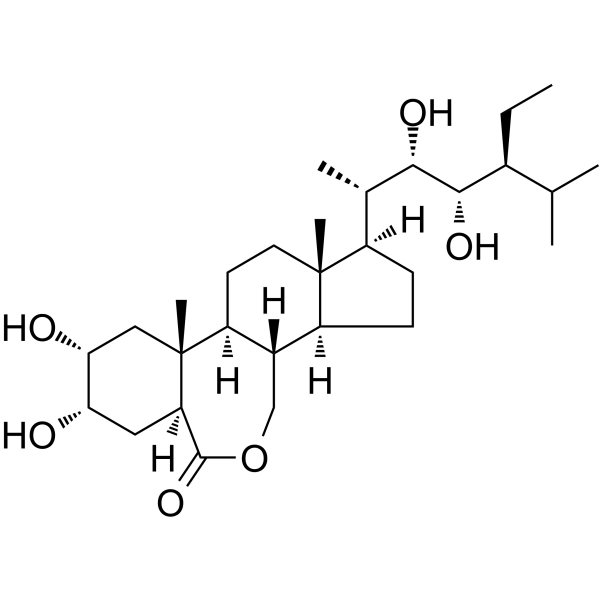 |
芸苔素内酯
CAS:80483-89-2 |
C29H50O6 |
|
Akt-dependent anabolic activity of natural and synthetic bra...
2011-06-23 [J. Med. Chem. 54 , 4057-4066, (2011)] |
|
Brassinosteroids protect Lycopersicon esculentum from cadmiu...
2010-03-01 [Protoplasma 239(1-4) , 3-14, (2010)] |
|
Synthesis and biological activity of furostanic analogues of...
2007-12-01 [Steroids 72(14) , 955-9, (2007)] |
|
Studies on steroidal plant-growth regulators (XVII)--a new s...
1991-06-01 [Sci. China,. Ser. B, Chem. Life Sci. Earth Sci. 34(6) , 706-11, (1991)] |
|
Induction of hexokinase I expression in normal and diabetic ...
2010-09-11 [Eur. J. Pharm. Sci. 41(1) , 1-9, (2010)] |