7-羟基-3-(4-甲氧基苯氧基)苯并吡喃-4-酮
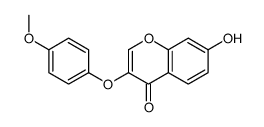
7-羟基-3-(4-甲氧基苯氧基)苯并吡喃-4-酮结构式

|
常用名 | 7-羟基-3-(4-甲氧基苯氧基)苯并吡喃-4-酮 | 英文名 | 7-hydroxy-3-(4-methoxyphenoxy)chromen-4-one |
|---|---|---|---|---|
| CAS号 | 137987-99-6 | 分子量 | 284.26300 | |
| 密度 | 1.373g/cm3 | 沸点 | 465.6ºC at 760 mmHg | |
| 分子式 | C16H12O5 | 熔点 | N/A | |
| MSDS | N/A | 闪点 | 176ºC |
| 中文名 | 7-羟基-3-(4-甲氧基苯氧基)苯并吡喃-4-酮 |
|---|---|
| 英文名 | 7-hydroxy-3-(4-methoxyphenoxy)chromen-4-one |
| 英文别名 | 更多 |
| 密度 | 1.373g/cm3 |
|---|---|
| 沸点 | 465.6ºC at 760 mmHg |
| 分子式 | C16H12O5 |
| 分子量 | 284.26300 |
| 闪点 | 176ºC |
| 精确质量 | 284.06800 |
| PSA | 68.90000 |
| LogP | 3.29950 |
| InChIKey | FUGYQGXYGJQPQY-UHFFFAOYSA-N |
| SMILES | COc1ccc(Oc2coc3cc(O)ccc3c2=O)cc1 |
| 蒸汽压 | 2.73E-09mmHg at 25°C |
| 折射率 | 1.64 |
|
~94% 
7-羟基-3-(4-甲氧基苯氧... 137987-99-6 |
| 文献:Vasil'ev, S. A.; Boyarchuk, V. L.; Luk'yanchikov, M. S.; Khilya, V. P. Pharmaceutical Chemistry Journal, 1991 , vol. 25, # 11 p. 816 - 821 Khimiko-Farmatsevticheskii Zhurnal, 1991 , vol. 25, # 11 p. 50 - 55 |
|
~% 
7-羟基-3-(4-甲氧基苯氧... 137987-99-6 |
| 文献:Chemistry of Heterocyclic Compounds (New York, NY, United States), , vol. 28, # 5 p. 497 - 502 Khimiya Geterotsiklicheskikh Soedinenii, Sbornik, , # 5 p. 595 - 600 |
|
~% 
7-羟基-3-(4-甲氧基苯氧... 137987-99-6 |
| 文献:Pharmaceutical Chemistry Journal, , vol. 25, # 11 p. 816 - 821 Khimiko-Farmatsevticheskii Zhurnal, , vol. 25, # 11 p. 50 - 55 |
|
~% 
7-羟基-3-(4-甲氧基苯氧... 137987-99-6 |
| 文献:Pharmaceutical Chemistry Journal, , vol. 25, # 11 p. 816 - 821 Khimiko-Farmatsevticheskii Zhurnal, , vol. 25, # 11 p. 50 - 55 |
| 7-羟基-3-(4-甲氧基苯氧基)苯并吡喃-4-酮上游产品 4 | |
|---|---|
| 7-羟基-3-(4-甲氧基苯氧基)苯并吡喃-4-酮下游产品 1 | |
|
实验名称:Primary cell-based high-throughput screening assay for identification of compounds th...
来源:Johns Hopkins Ion Channel Center
靶标:regulator of G-protein signaling 4 isoform 2 [Homo sapiens]
External Id:JHICC_RGS_Act_HTS
|
|
实验名称:Luminescence-based cell-based primary high throughput screening assay to identify ago...
来源:The Scripps Research Institute Molecular Screening Center
靶标:mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id:OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
实验名称:QFRET-based biochemical primary high throughput screening assay to identify exosite i...
来源:The Scripps Research Institute Molecular Screening Center
靶标:disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id:ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
实验名称:Fluorescence-based cell-based primary high throughput screening assay to identify ago...
来源:The Scripps Research Institute Molecular Screening Center
靶标:muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id:CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
实验名称:uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
来源:Burnham Center for Chemical Genomics
靶标:N/A
External Id:BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
实验名称:Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
来源:Broad Institute
靶标:FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id:2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
实验名称:Fluorescence-based cell-based primary high throughput screening assay to identify pos...
来源:The Scripps Research Institute Molecular Screening Center
靶标:muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id:CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
实验名称:Fluorescence polarization-based biochemical high throughput primary assay to identify...
来源:The Scripps Research Institute Molecular Screening Center
靶标:RecName: Full=Sialate O-acetylesterase; AltName: Full=H-Lse; AltName: Full=Sialic acid-specific 9-O-acetylesterase; Flags: Precursor [Homo sapiens]
External Id:SIAE_INH_FP_1536_1X%INH PRUN
|
|
实验名称:uHTS identification of small molecule modulators of NR3A
来源:Burnham Center for Chemical Genomics
靶标:N/A
External Id:SBCCG-A1015-NR3A-Primary-Assay
|
|
实验名称:Cell survival assay for modulators of telomere damage signalling
来源:15378
靶标:N/A
External Id:TELO_02
|
| 7-Hydroxy-3-(4-methoxyphenoxy)-4H-1-benzopyran-4-one |
| 7-Hydroxy-3-(4-methoxy-phenoxy)-chromen-4-one |
| 4H-1-Benzopyran-4-one,7-hydroxy-3-(4-methoxyphenoxy) |
![[3-(4-methoxyphenoxy)-4-oxochromen-7-yl] acetate结构式](https://image.chemsrc.com/caspic/221/137988-15-9.png) CAS号137988-15-9
CAS号137988-15-9





