3-(1H-苯并[d]咪唑-2-基)苯酚
![3-(1H-苯并[d]咪唑-2-基)苯酚结构式](https://image.chemsrc.com/caspic/143/6616-44-0.png)
3-(1H-苯并[d]咪唑-2-基)苯酚结构式

|
常用名 | 3-(1H-苯并[d]咪唑-2-基)苯酚 | 英文名 | 3-(1H-BENZOIMIDAZOL-2-YL)-PHENOL |
|---|---|---|---|---|
| CAS号 | 6616-44-0 | 分子量 | 210.23100 | |
| 密度 | N/A | 沸点 | N/A | |
| 分子式 | C13H10N2O | 熔点 | N/A | |
| MSDS | N/A | 闪点 | N/A |
| 中文名 | 3-(1H-苯并[d]咪唑-2-基)苯酚 |
|---|---|
| 英文名 | 3-(1H-benzimidazol-2-yl)phenol |
| 英文别名 | 更多 |
| 分子式 | C13H10N2O |
|---|---|
| 分子量 | 210.23100 |
| 精确质量 | 210.07900 |
| PSA | 48.91000 |
| LogP | 2.93550 |
| InChIKey | UDVFXRGKRCNQBH-UHFFFAOYSA-N |
| SMILES | Oc1cccc(-c2nc3ccccc3[nH]2)c1 |
| 海关编码 | 2933990090 |
|---|
|
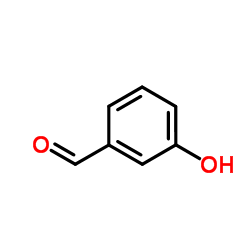
~94% ![3-(1H-苯并[d]咪唑-2-基)苯酚结构式](https://image.chemsrc.com/caspic/143/6616-44-0.png)
3-(1H-苯并[d]咪唑-2-基)苯酚 6616-44-0 |
| 文献:Zolfigol, Mohammad Ali; Khakyzadeh, Vahid; Moosavi-Zare, Ahmad Reza; Zare, Abdolkarim; Arghavani-Hadi, Parastoo; Mohammadi, Zahra; Beyzavi, Mohammad Hassan South African Journal of Chemistry, 2012 , vol. 65, p. 280 - 285 |
|
~82% ![3-(1H-苯并[d]咪唑-2-基)苯酚结构式](https://image.chemsrc.com/caspic/143/6616-44-0.png)
3-(1H-苯并[d]咪唑-2-基)苯酚 6616-44-0 |
| 文献:Weires, Nicholas A.; Boster, Jared; Magolan, Jakob European Journal of Organic Chemistry, 2012 , # 33 p. 6508 - 6512 |
|
~90% ![3-(1H-苯并[d]咪唑-2-基)苯酚结构式](https://image.chemsrc.com/caspic/143/6616-44-0.png)
3-(1H-苯并[d]咪唑-2-基)苯酚 6616-44-0 |
| 文献:Khalili, Saeed Bahadori; Sardarian, Ali Reza Monatshefte fur Chemie, 2012 , vol. 143, # 5 p. 841 - 846 |
|
~60% ![3-(1H-苯并[d]咪唑-2-基)苯酚结构式](https://image.chemsrc.com/caspic/143/6616-44-0.png)
3-(1H-苯并[d]咪唑-2-基)苯酚 6616-44-0 |
| 文献:Latif, N.; Mishriky, N.; Assad, F. M.; Meguid, S. Abdel Indian Journal of Chemistry, Section B: Organic Chemistry Including Medicinal Chemistry, 1982 , vol. 21, # 9 p. 872 - 874 |
|
~% ![3-(1H-苯并[d]咪唑-2-基)苯酚结构式](https://image.chemsrc.com/caspic/143/6616-44-0.png)
3-(1H-苯并[d]咪唑-2-基)苯酚 6616-44-0 |
| 文献:Latif, Nazih; Assaad, Fahmy M.; Hosni, Hanaa Liebigs Annalen der Chemie, 1987 , p. 495 - 498 |
|
~% ![3-(1H-苯并[d]咪唑-2-基)苯酚结构式](https://image.chemsrc.com/caspic/143/6616-44-0.png)
3-(1H-苯并[d]咪唑-2-基)苯酚 6616-44-0 |
| 文献:Latif, N.; Mishriky, N.; Assad, F. M.; Meguid, S. Abdel Indian Journal of Chemistry, Section B: Organic Chemistry Including Medicinal Chemistry, 1982 , vol. 21, # 9 p. 872 - 874 |
|
~% ![3-(1H-苯并[d]咪唑-2-基)苯酚结构式](https://image.chemsrc.com/caspic/143/6616-44-0.png)
3-(1H-苯并[d]咪唑-2-基)苯酚 6616-44-0 |
| 文献:Latif, Nazih; Assaad, Fahmy M.; Hosni, Hanaa Liebigs Annalen der Chemie, 1987 , p. 495 - 498 |
| 3-(1H-苯并[d]咪唑-2-基)苯酚上游产品 6 | |
|---|---|
| 3-(1H-苯并[d]咪唑-2-基)苯酚下游产品 0 | |
| 海关编码 | 2933990090 |
|---|---|
| 中文概述 | 2933990090. 其他仅含氮杂原子的杂环化合物. 增值税率:17.0%. 退税率:13.0%. 监管条件:无. 最惠国关税:6.5%. 普通关税:20.0% |
| 申报要素 | 品名, 成分含量, 用途, 乌洛托品请注明外观, 6-己内酰胺请注明外观, 签约日期 |
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
实验名称:Discovery of Small Molecules to Inhibit Human Cytomegalovirus Nuclear Egress
来源:ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
靶标:HCMV UL50
External Id:HMS1262
|
|
实验名称:Antioxidant activity assessed as superoxide radical anion scavenging activity by spec...
来源:ChEMBL
靶标:NON-PROTEIN TARGET
External Id:CHEMBL2400449
|
|
实验名称:Antioxidant activity assessed as hydroxyl radical scavenging activity after 1 hr by s...
来源:ChEMBL
靶标:NON-PROTEIN TARGET
External Id:CHEMBL2400448
|
|
实验名称:Antioxidant activity assessed as DPPH radical scavenging activity after 30 mins by sp...
来源:ChEMBL
靶标:N/A
External Id:CHEMBL2400447
|
|
实验名称:Binding affinity to Plasmodium falciparum 3D7 AMA1 (104 to 438 residues) expressed in...
来源:ChEMBL
靶标:N/A
External Id:CHEMBL4838835
|
|
实验名称:Cell-based high throughput primary assay to identify activators of GPR151
来源:The Scripps Research Institute Molecular Screening Center
靶标:RecName: Full=G-protein coupled receptor 151; AltName: Full=G-protein coupled receptor PGR7; AltName: Full=GPCR-2037; AltName: Full=Galanin receptor 4; AltName: Full=Galanin-receptor-like protein; Short=GalRL
External Id:GPR151_PHUNTER_AG_LUMI_1536_1X%ACT
|
|
实验名称:Antibacterial activity against Escherichia coli after 24 hrs by agar dilution method
来源:ChEMBL
靶标:Escherichia coli
External Id:CHEMBL2399645
|
|
实验名称:Antibacterial activity against Pseudomonas aeruginosa after 24 hrs by agar dilution m...
来源:ChEMBL
靶标:Pseudomonas aeruginosa
External Id:CHEMBL2399644
|
|
实验名称:Antibacterial activity against Staphylococcus aureus after 24 hrs by agar dilution me...
来源:ChEMBL
靶标:Staphylococcus aureus
External Id:CHEMBL2399643
|
|
实验名称:Antibacterial activity against Staphylococcus albus after 24 hrs by agar dilution met...
来源:ChEMBL
靶标:Staphylococcus epidermidis
External Id:CHEMBL2399642
|
| 2-<3-Oxy-phenyl>-benzimidazol |
| 3-benzimidazol-2-ylphenol |
| 2-<3-Hydroxy-phenyl>-benzimidazol |