| Structure | Name/CAS No. | Articles |
|---|---|---|
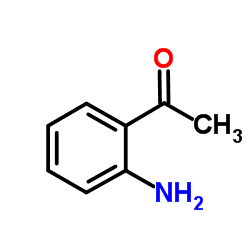 |
2'-Aminoacetophenone
CAS:551-93-9 |
|
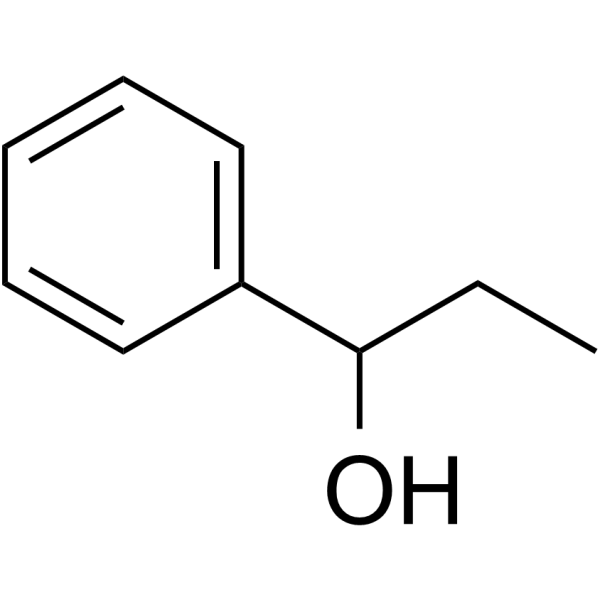 |
phenyl propanol
CAS:93-54-9 |
|
 |
2'-aminoacetophenone hydrochloride
CAS:25384-14-9 |