| Structure | Name/CAS No. | Articles |
|---|---|---|
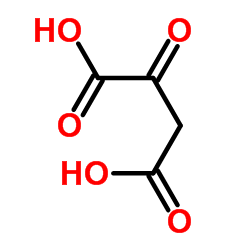 |
Oxaloacetic acid
CAS:328-42-7 |
|
 |
Phosphoenolpyruvate carboxylase
CAS:9067-77-0 |