Structural elucidation of metabolites of ginkgolic acid in rat liver microsomes by ultra-performance liquid chromatography/electrospray ionization tandem mass spectrometry and hydrogen/deuterium exchange.
Z H Liu, J Chen, L S Yu, H D Jiang, T W Yao, S Zeng
Index: Rapid Commun. Mass Spectrom. 23(13) , 1899-906, (2009)
Full Text: HTML
Abstract
Ginkgolic acids have been shown to possess allergenic as well as genotoxic and cytotoxic properties. The question arises whether the metabolism of ginkgolic acids in the liver could decrease or increase their toxicity. In this study, the in vitro metabolism of ginkgolic acid (15:1, GA), one component of ginkgo acids, was investigated as a model compound in Sprague-Dawley rat liver microsomes. The metabolites were analyzed by ultra-performance liquid chromatography coupled with photodiode array detector/negative-ion electrospray ionization tandem mass spectrometry (UPLC-PDA/ESI-MS/MS) and hydrogen/deuterium (H/D) exchange. The result showed that the benzene ring remained unchanged and the oxidations occurred at the side alkyl chain in rat liver microsomes. At least eight metabolites were found. Among them, six phase I metabolites were tentatively identified. This study might be useful for the investigation of toxicological mechanism of ginkgolic acids.Copyright (c) 2009 John Wiley & Sons, Ltd.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
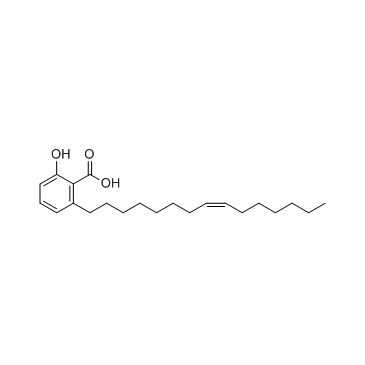 |
Ginkgolic acid C15:1
CAS:22910-60-7 |
C22H34O3 |
|
Design and evaluation of anacardic acid derivatives as antic...
2008-06-01 [Eur. J. Med. Chem. 43 , 1315-20, (2008)] |
|
Cytotoxicity of ginkgolic acid in HepG2 cells and primary ra...
2009-06-22 [Toxicol. Lett. 187(3) , 131-6, (2009)] |
|
Identification of TRIML2, a novel p53 target, that enhances ...
2015-02-01 [Mol. Cancer Res. 13(2) , 250-62, (2015)] |
|
Simultaneous quantification of flavonol glycosides, terpene ...
2013-01-01 [Biomed Res. Int. 2013 , 582591, (2013)] |
|
[Studies on supercritical CO2 fluid extraction for ginkgolic...
2003-06-01 [Zhong Yao Cai 26(6) , 428-9, (2003)] |