| Structure | Name/CAS No. | Articles |
|---|---|---|
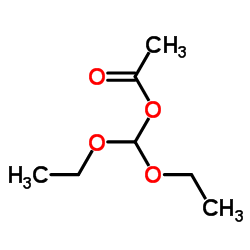 |
Diethoxymethyl acetate
CAS:14036-06-7 |
| Structure | Name/CAS No. | Articles |
|---|---|---|
 |
Diethoxymethyl acetate
CAS:14036-06-7 |