Synthesis of N-acetylglucosaminides of unconjugated and conjugated bile acids.
T Niwa, T Koshiyama, J Goto, T Nambara
Index: Steroids 57(11) , 522-9, (1992)
Full Text: HTML
Abstract
3-N-Acetylglucosaminides of unconjugated, glycine- and taurine-conjugated bile acids have been synthesized. Bile acids appropriately protected were condensed with acetochloroglucosamine through the 3 alpha-hydroxyl group by means of the Koenigs-Knorr reaction using cadmium carbonate as a catalyst. Subsequent borohydride reduction and/or alkaline hydrolysis provided desired 3-N-acetylglucosaminides of unconjugated bile acids. Glycine-conjugates were obtained from N-acetylglucosaminides of unconjugated bile acids and ethyl glycinate by the carbodiimide method. The preparation of N-acetylglucosaminides of taurine-conjugates was attained by the Koenigs-Knorr reaction of bile acid p-nitrophenyl esters followed by condensation with taurine. 7-N-Acetylglucosaminides of ursodeoxycholates were prepared in a similar fashion. The convenient synthesis of 3-N-acetylglucosaminides of unconjugated bile acids is also described.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
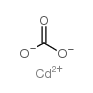 |
cadmium carbonate
CAS:513-78-0 |
CCdO3 |
|
Macromolecule oxidation and DNA repair in mussel (Mytilus ed...
2007-04-20 [Aquat. Toxicol. 82(1) , 27-35, (2007)] |
|
Heavy-metal remediation by a fungus as a means of production...
2005-08-02 [Langmuir 21(16) , 7220-4, (2005)] |
|
Top-down solid-phase fabrication of nanoporous cadmium oxide...
2007-02-28 [J. Am. Chem. Soc. 129(8) , 2333-7, (2007)] |
|
[Micromethod for the determination of polymorphonuclear gran...
1983-02-01 [Z. Gesamte Hyg. 29(2) , 69-70, (1983)] |
|
Synthesis of 15 alpha-hydroxyestrogen 15-N-acetylglucosamini...
1995-03-01 [Steroids 60(3) , 277-84, (1995)] |