| Structure | Name/CAS No. | Articles |
|---|---|---|
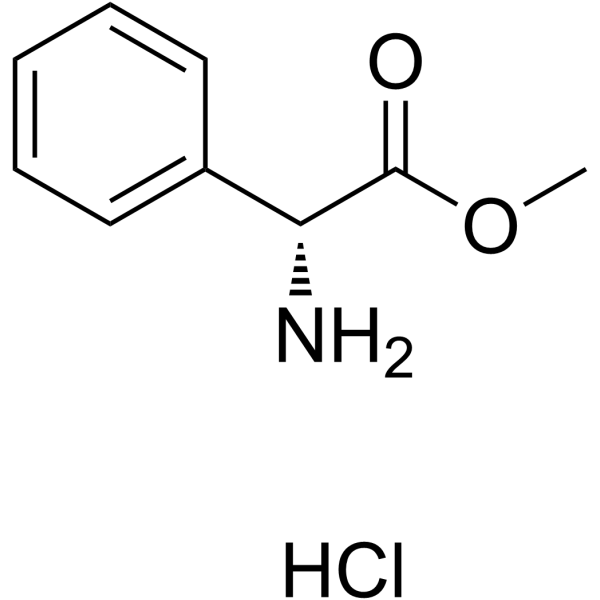 |
H-D-Phg-OMe.HCl
CAS:19883-41-1 |
|
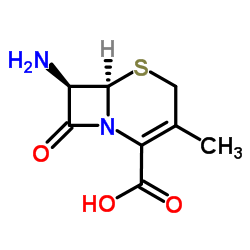 |
7-Aminodeacetoxycephalosporanic acid
CAS:22252-43-3 |