| Structure | Name/CAS No. | Articles |
|---|---|---|
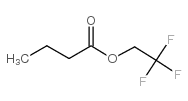 |
2,2,2-trifluoroethyl butyrate
CAS:371-27-7 |
|
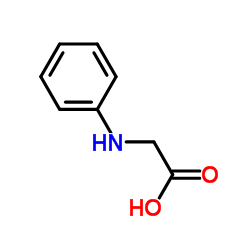 |
N-Phenylglycine
CAS:103-01-5 |
|
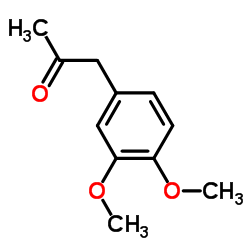 |
3,4-Dimethoxyphenylacetone
CAS:776-99-8 |