Mass spectrometry of underivatized 15-hydroxyeicosatetraenoic acid and 15-hydroxyeicosapentaenoic acid.
K Fogh, K Kragballe, E Larsen, H Egsgaard, V K Shukla
Index: Biomed. Environ. Mass Spectrom. 17(6) , 459-61, (1988)
Full Text: HTML
Abstract
A direct mass spectrometric method for 15-hydroxy metabolites of arachidonic acid and of eicosapentaenoic acid is described. 15-Hydroxyeicosanoids have the capacity to inhibit the formation of leukotrienes, potent mediators of inflammation formed by the 5-lipoxygenase pathway of arachidonic acid metabolism and may therefore have anti-inflammatory properties. 15-Hydroxyeicosatetraenoic acid (15-HETE) and 15-hydroxyeicosapentaenoic acid (15-HEPE) were synthesized using soybean lipoxygenase and arachidonic acid and eicosapentaenoic acid as substrates. These hydroxy fatty acids were then purified by reversed-phase high-performance liquid chromatography. This modified procedure provides rapid synthesis of multimilligram quantities of 15-hydroxyeicosanoids for in vitro and in vivo studies. Electron impact mass spectra of 15-HETE and 15-HEPE could be obtained directly without derivatizations.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
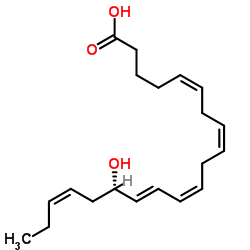 |
15-HEPE
CAS:86282-92-0 |
C20H30O3 |
|
Normalisation genes for expression analyses in the brown alg...
2008-01-01 [BMC Mol. Biol. 9 , 75, (2008)] |
|
[Endogenous ligands for PPARs].
2005-04-01 [Nihon Rinsho. 63(4) , 578-83, (2005)] |
|
Novel functional sets of lipid-derived mediators with antiin...
2000-10-16 [J. Exp. Med. 192(8) , 1197-204, (2000)] |
|
Involvement of 15-lipoxygenase in the inflammatory arthritis...
2012-07-01 [J. Cell. Biochem. 113(7) , 2279-89, (2012)] |
|
15-Hydroperoxyeicosapentaenoic acid inhibits arachidonic aci...
1996-05-20 [Biochim. Biophys. Acta 1300(3) , 171-6, (1996)] |