| Structure | Name/CAS No. | Articles |
|---|---|---|
 |
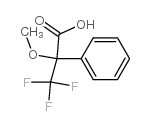
Mosher's acid
CAS:17257-71-5 |
|
 |
2,3-DICHLOROBENZALDOXIME
CAS:56135-03-6 |
|
 |
(R)-(+)-Mosher's acid
CAS:20445-31-2 |