| Structure | Name/CAS No. | Articles |
|---|---|---|
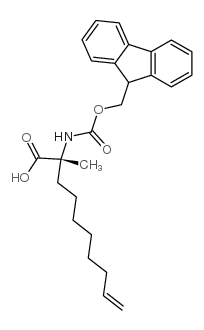 |
(R)-N-Fmoc-2-(7'-octenyl)alanine
CAS:945212-26-0 |
|
 |
Fmoc-(S)-2-(2-propenyl)Ala-OH
CAS:288617-71-0 |