Pharmacological characterization of the selective nonpeptide neuropeptide Y Y1 receptor antagonist BIBP 3226.
H N Doods, W Wienen, M Entzeroth, K Rudolf, W Eberlein, W Engel, H A Wieland
Index: J. Pharmacol. Exp. Ther. 275 , 136-142, (1995)
Full Text: HTML
Abstract
The present study was undertaken to investigate the in vitro and in vivo pharmacological profile of the novel, nonpeptide neuropeptide Y (NPY) Y1-selective antagonist, BIBP 3226 [(R)-N2-(diphenylacetyl)-N-[(4-hydroxyphenyl)methyl]-D-arginine-am ide], and a recently described peptidic structure [Ile-Glu-Pro-Orn-Tyr-Arg-Leu-Arg-Tyr-NH2, cyclic (2,4'), (2',4)-diamide]. BIBP 3226 antagonized the NPY Y1 receptor-mediated decrease in the twitch response in the rabbit vas deferens preparation with a pKb value of 6.98 +/- 0.06 (n = 16). It showed no affinity (EC50 > 1 microM) for NPY Y2 receptors in the rat vas deferens. NPY-induced increases in perfusion pressure in the isolated perfused rat kidney and rabbit ear preparations were antagonized with IC50 values of 26.8 +/- 4.5 (n = 4) and 214 +/- 30 nM (n = 4), respectively. The NPY-mediated potentiation of the noradrenaline elicited increase in perfusion pressure in the rat mesenteric bed was antagonized with an IC50 value of 976 (542-1760) nM. The NPY-induced increase in blood pressure in the pithed rat was inhibited by BIBP 3226 dose-dependently (ED50 = 0.11 +/- 0.03 mg/kg i.v.), whereas no effect of BIBP 3226 (1 mg/kg i.v.) was observed for the noradrenaline-, angiotensin-, endothelin- or vasopressin-induced pressor response. The data presented demonstrate that BIBP 3226 is a competitive and NPY Y1-selective antagonist. The peptidic compound proved to possess high potency for NPY Y1 receptors, but showed both agonistic as well as antagonistic properties.(ABSTRACT TRUNCATED AT 250 WORDS)
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
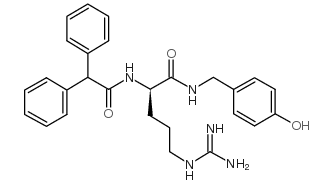 |
BIBP3226 trifluoroacetate salt
CAS:159013-54-4 |
C27H31N5O3 |
|
Frequency-dependent, cell type-divergent signaling in the hi...
2014-08-27 [J. Neurosci. 34(35) , 11769-80, (2014)] |
|
Selective inhibition of neuropeptide Y Y1 receptors by BIBP3...
1996-08-22 [Eur. J. Pharmacol. 310 , 55-60, (1996)] |
|
Non-competitive binding of the nonpeptide antagonist BIBP322...
1997-07-23 [Eur. J. Pharmacol. 331 , 275-284, (1997)] |
|
Involvement of amygdaloid neuropeptide Y in the anxiolytic e...
2014-05-01 [Neurosci. Lett. 567 , 19-23, (2014)] |