Tesmilifene may enhance breast cancer chemotherapy by killing a clone of aggressive, multi-drug resistant cells through its action on the p-glycoprotein pump.
Mark Vincent
Index: Med. Hypotheses 66(4) , 715-31, (2006)
Full Text: HTML
Abstract
Tesmilifene is a novel potentiator of chemotherapy which, when added to doxorubicin, achieved an unexpected and very large survival advantage over doxorubicin alone in a randomized trial in advanced breast cancer. This trial was unusual in that the early endpoints (response rate and median progression-free survival) were equivalent in the two arms, despite the ultimate survival difference. These aspects, coupled with the absence of a coherent molecular mechanism of action, and a pending confirmatory trial, have led oncologists to hold judgement on this drug. This paper reacts to this in three ways: firstly, a forensic subgroup analysis is presented with an explanation as to why it strongly supports the veracity of the survival difference; secondly a novel cellular explanation is provided for the decoupling of the early and late (survival) endpoints; finally, a molecular mechanism of action is proposed, for the first time, which reconciles the peculiarities of the trial with the laboratory data and background literature. This hypothesis explains how tesmilifene could meld two of the apparent strengths of the cancer cell (drug resistance pumps, and hypoxia-adapted energetics) into a potent weapon of self-destruction. Tesmilifene is proposed to allow chemotherapy (e.g. anthracycline or taxane) to additionally kill a small but critical population (clone) of aggressive, multi-drug resistant cells, the benefits of which cannot be appreciated until a period of time (about 6-8 months) has elapsed. These cells, present in women with more rapidly relapsing disease, very likely carry an energy-dependent extrusion pump which is paradoxically activated by tesmilifene plus the chemotherapy. The result is that, despite the chemotherapy's remaining extracellular, the cell dies from reactive oxygen species leaking from the electron chain transport in the abnormal mitochondria which characterize cancer. These mitochondria are activated in response to the ATP cost of this pump activation, in these predominantly glycolytic cells.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
 |
2-(4-Benzylphenoxy)-N,N-diethylethylamine hydrochloride
CAS:92981-78-7 |
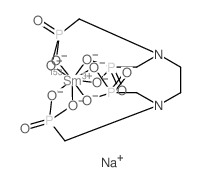
C6H12N2NaO12P4Sm---- |
|
Enhancement of antitumour activity of cisplatin by N,N-dieth...
1997-01-01 [Eur. J. Cancer 33(1) , 122-8, (1997)] |
|
Effect of alpha-FMH and DPPE on colony-forming properties of...
2002-07-01 [Curr. Med. Chem. 9(14) , 1349-57, (2002)] |
|
Effects of tesmilifene, a substrate of CYP3A and an inhibito...
2010-08-01 [J. Pharm. Pharmacol. 62(8) , 1084-8, (2010)] |
|
Effects of N,N-diethyl-2-[4-(phenylmethyl)phenoxy]ethanamine...
2000-01-03 [Eur. J. Pharmacol. 387(1) , 63-72, (2000)] |
|
Cerebral ischemia reperfusion-induced vasogenic brain edema ...
1998-08-01 [Eur. J. Pediatr. Surg. 8(4) , 216-9, (1998)] |