Iodine monochloride-amine complexes: an experimental and computational approach to new chiral electrophiles.
Jürgen Haas, Stewart Bissmire, Thomas Wirth
Index: Chemistry 11(19) , 5777-85, (2005)
Full Text: HTML
Abstract
Lactonizations are important steps in many synthetic sequences. Substrate-controlled reactions that use chiral auxiliaries or chiral alkenes have already been studied in depth. This study focuses on stereoselective reagent-controlled iodolactonizations, by application of a new method that uses complexes of iodine monochloride and various donor molecules. (R)-1,2,3,4-Tetrahydro-1-naphthylamine and other amines with similar structures were found to be efficient in the iodocyclization of 4-aryl-4-pentenoic acids. Calculations were performed on complexes of (R)-1,2,3,4-tetrahydro-1-naphthylamine with XCl (X = I, H) to identify possible reactive species in these iodocyclizations. Calculations were carried out at various levels of theory, including B3 LYP/6-31+G (d,p) by using a modified SDD basis set for iodine.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
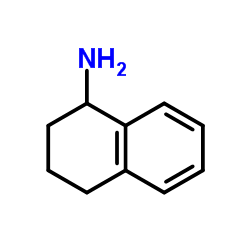 |
1,2,3,4-Tetrahydro-1-naphthylamine
CAS:2217-40-5 |
C10H13N |
|
Derivatization for the simultaneous LC/MS quantification of ...
2014-11-01 [J. Pharm. Biomed. Anal. 100 , 357-64, (2014)] |
|
Kinetic resolution of chiral amines with omega-transaminase ...
2001-05-05 [Biotechnol. Bioeng. 73(3) , 179-87, (2001)] |
|
Effects of isomers of apomorphines on dopamine receptors in ...
1985-09-16 [Life Sci. 37 , 1051, (1985)] |
|
Oxygen-18 studies on the oxidative deamination mechanism of ...
1982-05-01 [Arch. Biochem. Biophys. 215(2) , 433-43, (1982)] |
|
Rapid erasure of hippocampal memory following inhibition of ...
2016-01-01 [Nat. Commun. 7 , 10923, (2016)] |