| Structure | Name/CAS No. | Articles |
|---|---|---|
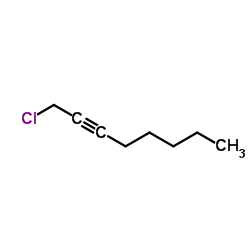 |
1-Chloro-2-octyne
CAS:51575-83-8 |
| Structure | Name/CAS No. | Articles |
|---|---|---|
 |
1-Chloro-2-octyne
CAS:51575-83-8 |