111790-37-5
| Name | 5-[(3aS,4S,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]-N-(2-aminoethyl)pentanamide |
|---|---|
| Synonyms |
biotinylethylenediamine
BAEA N-Biotinyl-ethylenediamine biotin ethylenediamine 1H-Thieno(3,4-d)imidazole-4-pentanamide,N-(2-aminoethyl)hexahydro-3-oxo-,(3aS,4S,6aR) N-[2-(D-biotinylamino)ethyl]amine N-(2-aminoethyl)biotinamide N-aminoethylaminobiotin [3aS-(3a|A,4|A,6a|A)]-N-(2-Aminoethyl)hexahydro-2-oxo-1H-thieno[3,4-d]imidazole-4-pentanamide (3aS,4S,6aR)-N-(2-Aminoethyl)hexahydro-2-oxo-1H-thieno[3,4-d]imidazole-4-pentanamide Biotinylamidoethylacetamide (3S,3a'S,6a'R)-N-Ethyl-2,2',8-trioxohexahydrospiro[azonane-3,4'-thieno[3,4-d]imidazole]-9-carboxamide N-biotinyl-1,2-diaminoethane |
| Description | Biotin-EDA is an alkyl chain-based PROTAC linker that can be used in the synthesis of PROTACs[1]. |
|---|---|
| Related Catalog | |
| Target |
Alkyl-Chain |
| In Vitro | PROTACs contain two different ligands connected by a linker; one is a ligand for an E3 ubiquitin ligase and the other is for the target protein. PROTACs exploit the intracellular ubiquitin-proteasome system to selectively degrade target proteins[1]. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 844.9±65.0 °C at 760 mmHg |
| Melting Point | 172-174ºC |
| Molecular Formula | C12H22N4O2S |
| Molecular Weight | 354.425 |
| Flash Point | 464.8±34.3 °C |
| Exact Mass | 354.136169 |
| PSA | 141.70000 |
| LogP | -0.77 |
| Vapour Pressure | 0.0±3.1 mmHg at 25°C |
| Index of Refraction | 1.612 |
|
~97% 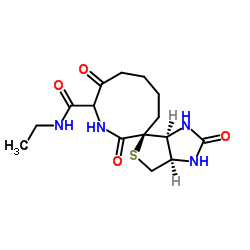
111790-37-5 |
| Literature: Yamada, Michio; Harada, Kenji; Maeda, Yutaka; Hasegawa, Tadashi New Journal of Chemistry, 2013 , vol. 37, # 11 p. 3762 - 3769 |
|
~99% 
111790-37-5 |
| Literature: Tao, Lei; Geng, Jin; Chen, Gaojian; Xu, Yingjian; Ladmiral, Vincent; Mantovani, Giuseppe; Haddleton, David. M. Chemical Communications, 2007 , # 33 p. 3441 - 3443 |
|
~% 
111790-37-5 |
| Literature: Bioorganic and medicinal chemistry, , vol. 11, # 2 p. 235 - 249 |
|
~% 
111790-37-5 |
| Literature: Angewandte Chemie - International Edition, , vol. 52, # 31 p. 8124 - 8128 Angew. Chem., , vol. 125, # 31 p. 8282 - 8286 |
|
~% 
111790-37-5 |
| Literature: Bioorganic and Medicinal Chemistry Letters, , vol. 20, # 14 p. 4077 - 4079 |
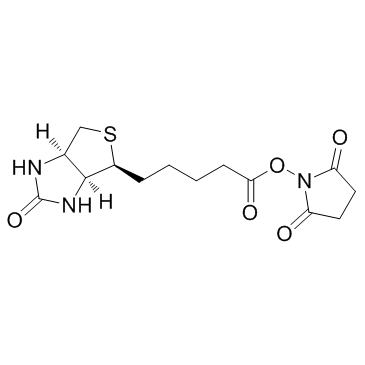
| Precursor 5 | |
|---|---|
| DownStream 1 | |



![N-[2-[[(2R)-2-amino-3-sulfanylpropanoyl]amino]ethyl]-5-[(4S)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanamide,2,2,2-trifluoroacetic acid structure](https://image.chemsrc.com/caspic/330/288144-42-3.png)