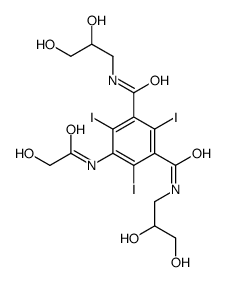
87771-40-2
| Name | 1-N,3-N-bis(2,3-dihydroxypropyl)-5-[(2-hydroxyacetyl)-(2-hydroxyethyl)amino]-2,4,6-triiodobenzene-1,3-dicarboxamide |
|---|---|
| Synonyms |
Ioversolum [Latin]
N,N'-bis(2,3-dihydroxypropyl)-5[(hydroxyacetyl)-(2-hydroxyethyl)-amino]-2,4,6-triiodo-1,3-benzenedicarboxamide N,N'-Bis(2,3-dihydroxypropyl)-5-[(hydroxyacetyl)(2-hydroxyethyl)amino]-2,4,6-triiodbenzol-1,3-dicarboxamid Optiray 160 UNII-N3RIB7X24K N,N'-bis(2,3-dihydroxypropyl)-5-[(hydroxyacétyl)(2-hydroxyéthyl)amino]-2,4,6-triiodobenzène-1,3-dicarboxamide Optiray N,N'-bis(2,3-dihydroxypropyl)-5-[(hydroxyacetyl)(2-hydroxyethyl)amino]-2,4,6-triiodobenzene-1,3-dicarboxamide MP-328 Optiray 320 [N,N'-bis-(2,3-dihydroxypropyl)-5-[(2-hydroxyacetyl)(2-hydroxyethyl)amino]-2,4,6-triiodoisophthalamide] Optiray 350 N,N'-bis(2,3-dihydroxypropyl)-5-[N-(2-hydroxyethyl)glycolamido]-2,4,6-triiodoisophthalamide Optiray 240 Ioversol MFCD00867959 N,N'-Bis(2,3-dihydroxypropyl)-5-[glycoloyl(2-hydroxyethyl)amino]-2,4,6-triiodoisophthalamide |
| Description | Ioversol is a nonionic iodinated contrast agent. |
|---|---|
| Related Catalog | |
| In Vitro | Ioversol (100 mg iodine/ml) exposure induces significantly increased lactate dehydrogenase release and decreased 3-(4,5-dimethyldiazol-2-yl)-2,5-diphenyl tetrazolium bromide conversion in NRK-52E cells. Ioversol significantly increases apoptosis and caspase-3 protein expression in the NRK-52E cells exposed to ioversol for 4 h. Ioversol treatment induces a significant increase in [Ca2+]i and intracellular ROS[1]. |
| In Vivo | In comparison with iothalamate, ioversol has a greatly reduced propensity to stimulate the release of endothelin, from cultured cells and when injected into anesthetized rats. Ioversol produces less renal vasoconstriction than does iothalamate, in control and in USIC rats, and the development of radiocontrast nephropathy, assessed by creatinine clearance and morphologic damage to the renal medulla, is largely avoided[2]. |
| Cell Assay | Cell viability is assessed by using the MTT uptake assay. The conversion of MTT, a tetrazolium salt, into formazan depends on the activity of a group of mitochondrial dehydrogenases and, thus, is an indicator of cell metabolic activity. The color of MTT tetrazole salt is measured with a spectrophotometer at the wavelength of 570 nm. Briefly, fresh MTT solution (10 mg/mL in 1× PBS) isadded (1:20 volume of medium) after removing the medium and the cells are incubated for 1.5 h. The cells are lysed and purple formazan dissolved into the solution by overnight incubation with MTT lysis buffer. The results are expressed as the percentage of undamaged control cells, assuming the absorbance of control wells as 100%. |
| References |
| Density | 2.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 864.9±65.0 °C at 760 mmHg |
| Melting Point | 180-182ºC |
| Molecular Formula | C18H24I3N3O9 |
| Molecular Weight | 807.111 |
| Flash Point | 476.9±34.3 °C |
| Exact Mass | 806.864624 |
| PSA | 199.89000 |
| LogP | -4.01 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.739 |
| Storage condition | Refrigerator |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RIDADR | NONH for all modes of transport |
|---|
|
~% 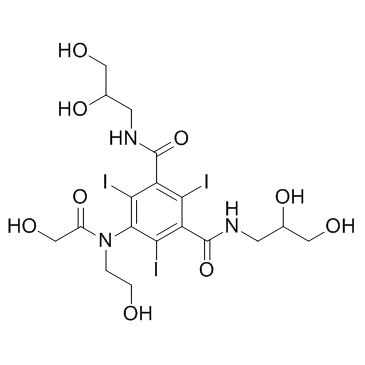
87771-40-2 |
| Literature: WO2008/104853 A1, ; Page/Page column 15; 11-12 ; |
| Precursor 2 | |
|---|---|
| DownStream 0 | |