50594-67-7
| Name | acifluorfen-methyl |
|---|---|
| Synonyms |
Methyl 5-[2-chloro-4-(trifluoromethyl)phenoxy]-2-nitrobenzoate
5-[2-Chloro-4-(trifluorométhyl)phénoxy]-2-nitrobenzoate de méthyle 5-[2-Chloro-4-(trifluoromethyl)phenoxy]-2-nitrobenzoic acid methyl ester Acifluorfen methyl ester Benzoic acid, 5-[2-chloro-4-(trifluoromethyl)phenoxy]-2-nitro-, methyl ester methyl 5-(2-chloro-α,α,α-trifluoro-p-tolyloxy)-2-nitrobenzoate Methyl-5-[2-chlor-4-(trifluormethyl)phenoxy]-2-nitrobenzoat Acifluorfen metabolite |
| Description | Acifluorfen-methyl is an inhibitor of the heme and chlorophyll biosynthetic enzyme protoporphyrinogen oxidase (Protox). Acifluorfen-methyl is a photobleaching herbicide[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.5±0.1 g/cm3 |
|---|---|
| Boiling Point | 392.3±42.0 °C at 760 mmHg |
| Molecular Formula | C15H9ClF3NO5 |
| Molecular Weight | 375.684 |
| Flash Point | 191.0±27.9 °C |
| Exact Mass | 375.012146 |
| PSA | 81.35000 |
| LogP | 4.32 |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.547 |
| HS Code | 2918990090 |
|---|
|
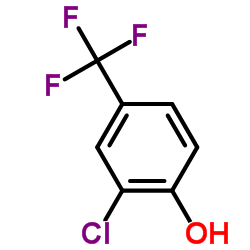
~27% 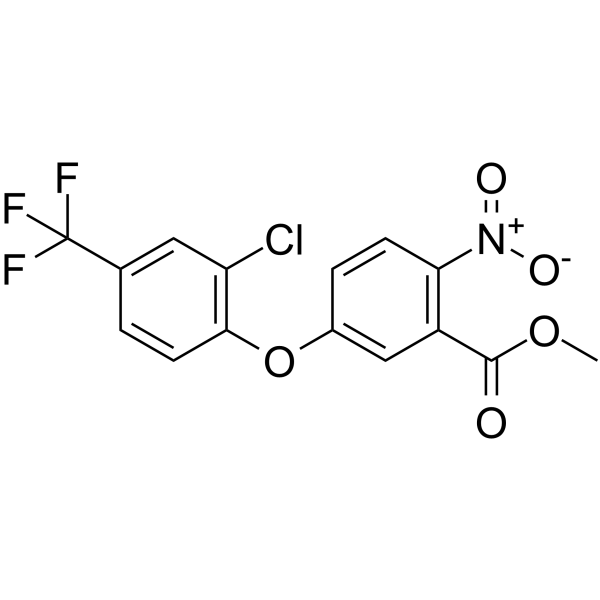
50594-67-7 |
| Literature: Rohm and Haas Company Patent: US4259510 A1, 1981 ; |
|
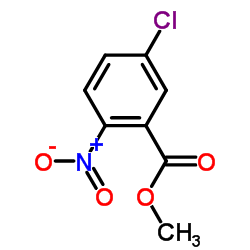
~82% 
50594-67-7 |
| Literature: Rohm and Haas Company Patent: US4259510 A1, 1981 ; |
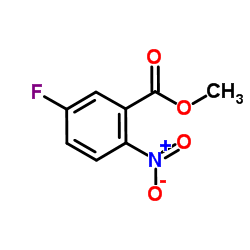
|
~% 
50594-67-7 |
| Literature: Rohm and Haas Company Patent: US4031131 A1, 1977 ; |
| Precursor 4 | |
|---|---|
| DownStream 2 | |
| HS Code | 2918990090 |
|---|---|
| Summary | 2918990090. other carboxylic acids with additional oxygen function and their anhydrides, halides, peroxides and peroxyacids; their halogenated, sulphonated, nitrated or nitrosated derivatives. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
![methyl 2-amino-5-[2-chloro-4-(trifluoromethyl)phenoxy]benzoate structure](https://image.chemsrc.com/caspic/226/58105-66-1.png)