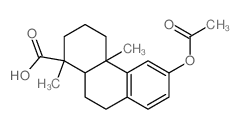
5947-49-9
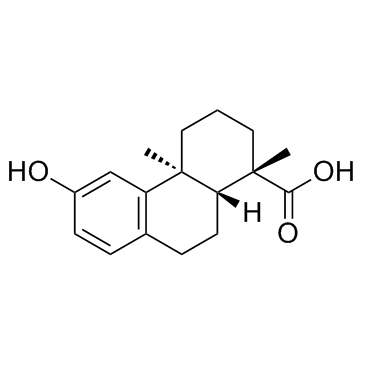
| Name | podocarpic acid |
|---|---|
| Synonyms |
1 4A-DIMETHYL-6-HYDROXY-1 2 3,4A 9 10 10A-OCTAHYDRO-1-PHENANTHRENECARBOXYLIC ACID
MFCD00074825 PODOCARPIC ACID Einecs 227-706-3 12-HYDROXYPODOCARPA-8,11,13-TRIEN-16-OIC ACID |
| Description | Podocarpic acid is a natural product, which has the best all-round positive effect and acts as a novel TRPA1 activator. |
|---|---|
| Related Catalog | |
| In Vitro | Podocarpic acid anhydride acts as a 1 nM agonist of LXRalpha and beta receptors. It shows over 8-10-fold better activator of LXR receptors compared to one of the natural ligands, 22-(R)-hydroxy cholesterol, in HEK-293 cells[2]. |
| In Vivo | Podocarpic acid activates SKN-1 in C. elegans, similar to known Nrf2 activators such as α-lipoic acid (LA). Podocarpic acid- or LA-induced SKN-1 activation also requires TRPodocarpic acid-1: trPodocarpic acid-1 knockdown in glod-4;gst-4p::gfp animals reduces expression of gst-4 to wild-type levels. A and LA supplementation results in a robust Ca2+ flux, which is significantly reduces when the Ca2+-impermeable TRPodocarpic acid-1E1018A channel is present, suggesting that TRPodocarpic acid-1 activation is key for these drugs' function. Finally, Podocarpic acid and LA alleviate the Podocarpic acidthogenic phenotypes of glod-4 animals by reverting the high endogenous MGO and GO to almost wild-type-like levels[1]. |
| References |
| Melting Point | 193-196 ℃(lit.) |
|---|---|
| Molecular Formula | C17H22O3 |
| Molecular Weight | 274.35500 |
| Exact Mass | 274.15700 |
| PSA | 57.53000 |
| LogP | 3.48710 |
| Storage condition | -20°C |
| HS Code | 2916209090 |
|---|
| Precursor 3 | |
|---|---|
| DownStream 4 | |
| HS Code | 2916209090 |
|---|---|
| Summary | 2916209090 other cyclanic, cyclenic or cyclotherpenic monocarboxylic acids, their anhydrides, halides, peroxides, peroxyacids and their derivatives VAT:17.0% Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) MFN tariff:6.5% General tariff:30.0% |
![(3S)-3-[(1R)-3-(p-methoxyphenyl)-1-(nitromethyl)propyl]-3-methyltetrahydropyran-2-one structure](https://image.chemsrc.com/caspic/152/123427-81-6.png)
![8-Methyl-[3]phenanthrol structure](https://image.chemsrc.com/caspic/093/111589-13-0.png)
![3a-hydroperoxy-6,9a-dimethyl-2-oxo-4,5,5a,7,8,9-hexahydrobenzo[e][1]benzofuran-6-carboxylic acid structure](https://image.chemsrc.com/caspic/295/81801-25-4.png)