122547-49-3
| Name | Faropenem sodium hydrate |
|---|---|
| Synonyms |
Natrium-(5R,6S)-6-[(1R)-1-hydroxyethyl]-7-oxo-3-[(2R)-tetrahydrofuran-2-yl]-4-thia-1-azabicyclo[3.2.0]hept-2-en-2-carboxylat
sodium (5R,6S)-6-[(1R)-1-hydroxyethyl]-7-oxo-3-[(2R)-tetrahydrofuran-2-yl]-4-thia-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylate 4-Thia-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid, 6-[(1R)-1-hydroxyethyl]-7-oxo-3-[(2R)-tetrahydro-2-furanyl]-, sodium salt, (5R,6S)- (1:1) Faropenem sodium 4-Thia-1-azabicyclo[3.2.0]hept-2-ene-3-carboxylic acid, 6-[(1R)-1-hydroxyethyl]-7-oxo-2-[(2R)-tetrahydro-2-furanyl]-, sodium salt, (5R,6S)- (1:1) Sodium (5R,6S)-6-[(1R)-1-hydroxyethyl]-7-oxo-2-[(2R)-tetrahydro-2-furanyl]-4-thia-1-azabicyclo[3.2.0]hept-2-ene-3-carboxylate Sodium (5R,6S)-6-[(1R)-1-hydroxyethyl]-7-oxo-3-[(2R)-tetrahydro-2-furanyl]-4-thia-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylate |
| Description | Faropenem sodium is an orally bioavailable penem antibiotic which can efficiently kill Mycobacterium tuberculosis. |
|---|---|
| Related Catalog | |
| Target |
Bacterial[1] |
| References |
| Boiling Point | 570.2ºC at 760 mmHg |
|---|---|
| Molecular Formula | C12H15NNaO5S |
| Molecular Weight | 308.31 |
| Flash Point | 298.7ºC |
| PSA | 115.20000 |
| Vapour Pressure | 2.23E-15mmHg at 25°C |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
|
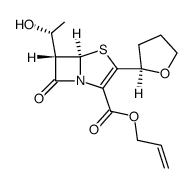
~87% 
122547-49-3 |
| Literature: Huang, Jian-Ping; Chen, Xu-Xiang; Gu, Shuang-Xi; Zhao, Lei; Chen, Wen-Xue; Chen, Fen-Er Organic Process Research and Development, 2010 , vol. 14, # 4 p. 939 - 941 |
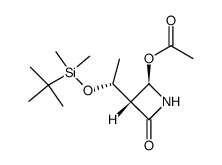
|
~% 
122547-49-3 |
| Literature: Bioorganic and Medicinal Chemistry, , vol. 5, # 7 p. 1389 - 1399 |
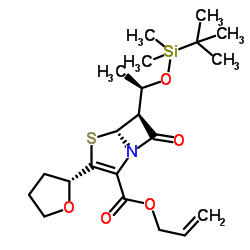
|
~% 
122547-49-3 |
| Literature: Bioorganic and Medicinal Chemistry, , vol. 5, # 7 p. 1389 - 1399 |
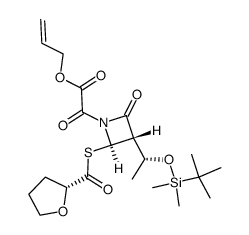
|
~% 
122547-49-3 |
| Literature: Bioorganic and Medicinal Chemistry, , vol. 5, # 7 p. 1389 - 1399 |
|
~% 
122547-49-3 |
| Literature: Bioorganic and Medicinal Chemistry, , vol. 5, # 7 p. 1389 - 1399 |
| Precursor 4 | |
|---|---|
| DownStream 0 | |