1-NITRO-2-NAPHTHALDEHYDE
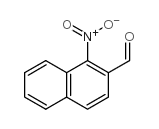
1-NITRO-2-NAPHTHALDEHYDE structure
|
Common Name | 1-NITRO-2-NAPHTHALDEHYDE | ||
|---|---|---|---|---|
| CAS Number | 101327-84-8 | Molecular Weight | 201.17800 | |
| Density | 1.368g/cm3 | Boiling Point | 384.6ºC at 760 mmHg | |
| Molecular Formula | C11H7NO3 | Melting Point | 109-110ºC(lit.) | |
| MSDS | Chinese USA | Flash Point | 197.5ºC | |
| Name | 1-nitronaphthalene-2-carbaldehyde |
|---|---|
| Synonym | More Synonyms |
| Density | 1.368g/cm3 |
|---|---|
| Boiling Point | 384.6ºC at 760 mmHg |
| Melting Point | 109-110ºC(lit.) |
| Molecular Formula | C11H7NO3 |
| Molecular Weight | 201.17800 |
| Flash Point | 197.5ºC |
| Exact Mass | 201.04300 |
| PSA | 62.89000 |
| LogP | 3.08370 |
| Vapour Pressure | 4.05E-06mmHg at 25°C |
| Index of Refraction | 1.708 |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| RIDADR | NONH for all modes of transport |
| HS Code | 2913000090 |
| HS Code | 2913000090 |
|---|---|
| Summary | HS: 2913000090 halogenated, sulphonated, nitrated or nitrosated derivatives of products of heading 2912 Educational tariff:17.0% Tax rebate rate:9.0% Regulatory conditions:none Most favored nation tariff:5.5% General tariff:30.0% |
|
The Baylis-Hillman acetates as a valuable source for one-pot multistep synthesis: a facile synthesis of functionalized tri-/tetracyclic frameworks containing azocine moiety.
Org. Lett. 9(13) , 2453-6, (2007) The Baylis-Hillman acetates have been conveniently transformed into tri-/tetracyclic heterocyclic frameworks containing an important azocine moiety via one-pot multistep protocol involving alkylation,... |
|
|
Materials and systems for two photon 3-D ROM devices. Dvornikov AS, et al.
IEEE Trans. Compon. Packag. Tech. 20(2) , 203-212, (1997)
|
| 1-nitro-2-naphthalenecarboxaldehyde |
| 1-nitro-[2]naphthaldehyde |
| 1-nitronaphthalene-2-carboxaldehyde |
| MFCD03427164 |
| 2-Naphthalenecarboxaldehyde,1-nitro |
| 1-nitro-2-naphthylaldehyde |
| 1-nitro-naphthalene-2-carbaldehyde |
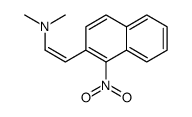 CAS#:176853-40-0
CAS#:176853-40-0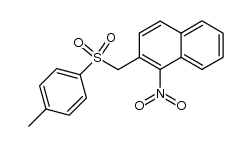 CAS#:136613-49-5
CAS#:136613-49-5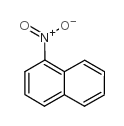 CAS#:86-57-7
CAS#:86-57-7 CAS#:881-03-8
CAS#:881-03-8![2-phenyl-1H-benzo[g]indole structure](https://www.chemsrc.com/caspic/329/33555-17-8.png) CAS#:33555-17-8
CAS#:33555-17-8 CAS#:176853-41-1
CAS#:176853-41-1![benzo[h]quinolin-2-amine structure](https://www.chemsrc.com/caspic/018/67410-22-4.png) CAS#:67410-22-4
CAS#:67410-22-4
