taprostene
Modify Date: 2025-08-25 15:42:44
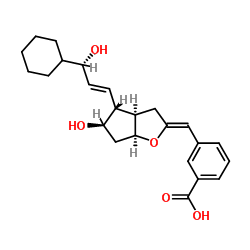
taprostene structure
|
Common Name | taprostene | ||
|---|---|---|---|---|
| CAS Number | 108945-35-3 | Molecular Weight | 398.492 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 609.4±55.0 °C at 760 mmHg | |
| Molecular Formula | C24H30O5 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 209.2±25.0 °C | |
Use of taprosteneTaprostene (CG-4203) is a synthetic, chemically stable analogue of Prostacyclin (PGI2). Taprostene exhibits endothelium and myocardial protecting actions after acute myocardial ischemia and reperfusion in cats. Taprostene enhances cytoprotective actions, while minimizing unwanted hemodynamic effects[1]. |
| Name | Taprostene |
|---|---|
| Synonym | More Synonyms |
| Description | Taprostene (CG-4203) is a synthetic, chemically stable analogue of Prostacyclin (PGI2). Taprostene exhibits endothelium and myocardial protecting actions after acute myocardial ischemia and reperfusion in cats. Taprostene enhances cytoprotective actions, while minimizing unwanted hemodynamic effects[1]. |
|---|---|
| Related Catalog | |
| Target |
Prostaglandin Receptor[1] |
| In Vivo | Taprostene (100 ng/kg/min) is infused intravenously starting 30 minutes postocclusion of the left anterior descending coronary artery followed by reperfusion 1 hour later in a 6-hour model of myocardial ischemia (MI) with reperfusion in anesthetized cats. Taprostene infusion results in significantly lower plasma creatine phosphokinase activities for the MI + Taprostene group compared with the MI + vehicle group.Taprostene has a profile of activity including prevention of aggregation in cat platelets15 at concentrations that are much lower than that required to produce significant vasodilator activity in rabbit aortic rings. In addition to antiaggregatory and cytoprotective effects in circulatory shock, Taprostene exerts beneficial effects in acute inflammatory states and in rat models of myocardial hypoxia and permanent ischemia.A variety of infusion rates of Taprostene from 50 to 200 ng/kg/min were initially used to obtain an infusion rate that produced minimal hemodynamic (i.e., vasodilator) effects but still exerted cardioprotective effects. Taprostene treatment inhibits neutrophils adhering to the myocardial endothelium in both jeopardized and necrotic myocardial tissue after ischemia and reperfusion[1]. Animal Model: Adult male cats (2.5-3.5 kg)[1] Dosage: 100 ng/kg Administration: Infused intravenously at a rate of 100 ng/kg/min until the end of the experiment (i.e., for 5.5 hours). Result: In six cat aortic rings, 1-100 ng/mL failed to exert any vasorelaxant effect. Relaxed the vascular rings 34% at 300 ng/mL. The EC50 was 520 ng/mL, a value 26 times that of its antiplatelet aggregatory effect in cat platelet-rich plasma. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 609.4±55.0 °C at 760 mmHg |
| Molecular Formula | C24H30O5 |
| Molecular Weight | 398.492 |
| Flash Point | 209.2±25.0 °C |
| Exact Mass | 398.209320 |
| PSA | 86.99000 |
| LogP | 3.78 |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.670 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| 3-[(Z)-{(3aR,4R,5R,6aS)-4-[(1E,3S)-3-Cyclohexyl-3-hydroxy-1-propen-1-yl]-5-hydroxyhexahydro-2H-cyclopenta[b]furan-2-ylidene}methyl]benzoic acid |
| [3aR-[2Z,3aa,4a(1E,3S*)-5b,6aa]]-3-[[4-(3-Cyclohexyl-3-hydroxy-1-propenyl)hexahydro-5-hydroxy-2H-cyclopenta[b]furan-2-ylidene]methyl]benzoic acid |
| a-[(2Z,3aR,4R,5R,6aS)-4-[(1E,3S)-3-Cyclohexyl-3-hydroxypropenyl]hexahydro-5-hydroxy-2H-cyclopenta[b]furan-2-ylidene]-m-toluic acid |
| [(5Z,13E,9a,11a,15S)-2,3,4-Trinor-1,5-inter-m-phenylene-6,9-epoxy-11,15-dihydroxy-15-cyclohexyl-16,17,18,19,20-pentanor]prosta-5,13-dienoic acid |
| Benzoic acid, 3-[(Z)-[(3aR,4R,5R,6aS)-4-[(1E,3S)-3-cyclohexyl-3-hydroxy-1-propen-1-yl]hexahydro-5-hydroxy-2H-cyclopenta[b]furan-2-ylidene]methyl]- |
| taprostene |