Abemaciclib(LY2835219)
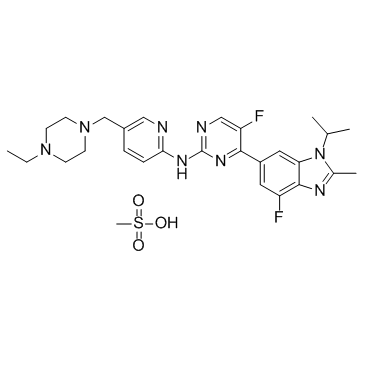
Abemaciclib(LY2835219) structure
|
Common Name | Abemaciclib(LY2835219) | ||
|---|---|---|---|---|
| CAS Number | 1231930-82-7 | Molecular Weight | 602.699 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C28H36F2N8O3S | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of Abemaciclib(LY2835219)Abemaciclib (LY2835219) (methanesulfonate) is a selective CDK4/6 inhibitor with IC50s of 2 nM and 10 nM for CDK4 and CDK6, respectively. |
| Name | N-[5-[(4-ethylpiperazin-1-yl)methyl]pyridin-2-yl]-5-fluoro-4-(7-fluoro-2-methyl-3-propan-2-ylbenzimidazol-5-yl)pyrimidin-2-amine,methanesulfonic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Abemaciclib (LY2835219) (methanesulfonate) is a selective CDK4/6 inhibitor with IC50s of 2 nM and 10 nM for CDK4 and CDK6, respectively. |
|---|---|
| Related Catalog | |
| Target |
Cdk4/cyclin D1:2 nM (IC50) CDK6/cyclinD1:10 nM (IC50) CDK2/cyclinE:504 nM (IC50) CDK9/cyclinT1:57 nM (IC50) CDK1/cyclinB1:1627 nM (IC50) CDK7/Mat1/cyclinH1:3910 nM (IC50) Cdk5/p25:355 nM (IC50) CDK5/p35:287 nM (IC50) PIM1:50 nM (IC50) PIM2:3400 nM (IC50) HIPK2:31 nM (IC50) DYRK2:61 nM (IC50) CK2:117 nM (IC50) GSK3b:192 nM (IC50) JNK3:389 nM (IC50) FLT3 (D835Y):403 nM (IC50) FLT3:3960 nM (IC50) DRAK1:659 nM (IC50) |
| In Vitro | Abemaciclib (LY2835219) reduces cell viability with the IC50 values ranging from 0.5 μM to 0.7 μM, inhibits Akt and ERK signaling but not mTOR activation at head and neck squamous cell carcinoma (HNSCC) cells[1]. Abemaciclib (LY2835219) shows inhibition on A375R1-4, M14R, and SH4R with EC50 values ranging from 0.3 to 0.6 μM; Abemaciclib inhibits the proliferation of the parental A375 and resistant A375RV1 and A375RV2 cells with similar potencies with IC50 values of 395, 260, and 463 nM, respectively[2]. Abemaciclib (LY2835219) inhibits CDK4 and CDK6 with low nanomolar potency, inhibits Rb phosphorylation resulting in a G1 arrest and inhibition of proliferation, and its activity is specific for Rb-proficient cells[3]. |
| In Vivo | Abemaciclib (LY2835219) (45 mg/kg, p.o.) in combination with everolimus causes a cooperative antitumor effect in HNSCC xenograft tumor[1]. Abemaciclib (LY2835219) (45 or 90 mg/kg, p.o.) shows significant tumor growth inhibition in an A375 xenograft model[2]. |
| Cell Assay | Cells are seeded in a 96-well plate, allowed to adhere overnight, and treated with DMSO control (0.1% v/v) or the indicated compounds for 72 h. Cell viability and proliferation are determined using a Cell Counting Kit according to the manufacturer's instructions. The interaction between Abemaciclib (LY2835219) and mTOR inhibitor is determined using CompuSyn. Combination index (CI) values of 1 indicates and additive drug interaction, whereas a CI of < 1 is synergistic and a CI of > 1 is antagonistic. |
| Animal Admin | Six-week-old BALB/c female nude mice are injected subcutaneously with OSC-19 (1×106) cells. When tumor sizes reach approximately 100 mm3, mice are randomized by tumor size and subjected to each treatment. At least 5 mice per treatment group are included. Each group of mice is dosed via daily oral gavage with vehicle, Abemaciclib (LY2835219) (45 mg/kg/d or 90 mg/kg/d), Everolimus (5 mg/kg/d), or a combination of both. The Abemaciclib (LY2835219) is dissolved in 1% HEC in 20 mM phosphate buffer (pH2.0). Tumor size and body weight are measured twice weekly. Tumor volumes are calculated using the following formula: V=(L × W2)/2 (L, Length; W, width). Mice are gavaged a final time on day 14 and sacrificed the following day. The tumors are removed for Western blot and immunohistochemistry. |
| References |
| Molecular Formula | C28H36F2N8O3S |
|---|---|
| Molecular Weight | 602.699 |
| Exact Mass | 602.259888 |
| PSA | 137.75000 |
| LogP | 5.47050 |
| InChIKey | NCJPFQPEVDHJAZ-UHFFFAOYSA-N |
| SMILES | CCN1CCN(Cc2ccc(Nc3ncc(F)c(-c4cc(F)c5nc(C)n(C(C)C)c5c4)n3)nc2)CC1.CS(=O)(=O)O |
|
Name: Thermal Shift Assay. Domain: start/stop: N44-E168
Source: ChEMBL
Target: Bromodomain-containing protein 4
External Id: CHEMBL5060944
|
|
Name: Thermal Shift Assay. Domain start/stop: M626-G740
Source: ChEMBL
Target: Peregrin
External Id: CHEMBL4650107
|
|
Name: Thermal Shift Assay. Domain: start/stop: M1-L298
Source: ChEMBL
Target: Cyclin-dependent kinase 2
External Id: CHEMBL5062802
|
|
Name: Thermal Shift Assay. Domain: start/stop: E122-S403
Source: ChEMBL
Target: Aurora kinase A
External Id: CHEMBL5067851
|
|
Name: Thermal Shift Assay. Domain: start/stop: M1-K294
Source: ChEMBL
Target: Casein kinase I isoform delta
External Id: CHEMBL5064135
|
|
Name: Thermal Shift Assay. Domain: start/stop: M26-R383
Source: ChEMBL
Target: Glycogen synthase kinase-3 beta
External Id: CHEMBL5065993
|
|
Name: Thermal Shift Assay. Domain: start/stop: S229-K512
Source: ChEMBL
Target: Tyrosine-protein kinase ABL1
External Id: CHEMBL5060419
|
|
Name: SARS-CoV-2 3CL-Pro protease inhibition percentage at 20µM by FRET kind of response f...
Source: ChEMBL
Target: Replicase polyprotein 1ab
External Id: CHEMBL4495582
|
|
Name: Thermal Shift Assay. Domain: start/stop: M626-G746
Source: ChEMBL
Target: Peregrin
External Id: CHEMBL5062277
|
|
Name: Inhibition of CDK2/Cyclin A2 (unknown origin) using FAM-labeled peptide as substrate ...
Source: ChEMBL
Target: Cyclin-A2
External Id: CHEMBL4275364
|
| N-{5-[(4-Ethyl-1-piperazinyl)methyl]-2-pyridinyl}-5-fluoro-4-(4-fluoro-1-isopropyl-2-methyl-1H-benzimidazol-6-yl)-2-pyrimidinamine methanesulfonate (1:1) |
| CS-1229 |
| QCR-274 |
| 2-Pyrimidinamine, N-[5-[(4-ethyl-1-piperazinyl)methyl]-2-pyridinyl]-5-fluoro-4-[4-fluoro-2-methyl-1-(1-methylethyl)-1H-benzimidazol-6-yl]-, methanesulfonate (1:1) |
| UNII-KKT462Q807 |
| N-(5-((4-ethylpiperazin-1-yl)methyl)pyridin-2-yl)-5-fluoro-4-(4-fluoro-1-isopropyl-2-methyl-1H-benzo[d]imidazol-6-yl)pyrimidin-2-amine methanesulfonate |
| LY2835219 |
| Abemaciclib mesylate |

