RifamycinS
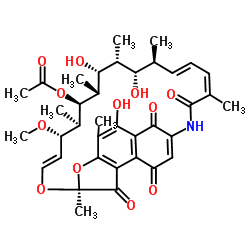
RifamycinS structure
|
Common Name | RifamycinS | ||
|---|---|---|---|---|
| CAS Number | 13553-79-2 | Molecular Weight | 695.753 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 917.4±65.0 °C at 760 mmHg | |
| Molecular Formula | C37H45NO12 | Melting Point | 179-181ºC (dec.) | |
| MSDS | N/A | Flash Point | 508.6±34.3 °C | |
Use of RifamycinSRifamycin S is a quinone and an antibiotic agnet against Gram-positive bacteria (including MRSA). Rifamycin S is the oxidized forms of a reversible oxidation-reduction system involving two electrons. Rifamycin S generates reactive oxygen species (ROS) and inhibits microsomal lipid peroxidation. Rifamycin S can be used for tuberculosis and leprosy[1][2][3]. |
| Name | Rifamycin S |
|---|---|
| Synonym | More Synonyms |
| Description | Rifamycin S is a quinone and an antibiotic agnet against Gram-positive bacteria (including MRSA). Rifamycin S is the oxidized forms of a reversible oxidation-reduction system involving two electrons. Rifamycin S generates reactive oxygen species (ROS) and inhibits microsomal lipid peroxidation. Rifamycin S can be used for tuberculosis and leprosy[1][2][3]. |
|---|---|
| Related Catalog | |
| Target |
Gram-positive bacteria[3] Reactive oxygen species (ROS)[1] |
| In Vitro | The inhibition of bacterial growth by Rifamycin SV is due to the production of active species of oxygen resulting from the oxidation-reduction cycle of Rifamycin SV in the cells. The aerobic oxidation of Rifamycin SV to Rifamycin S is induced by metal ions, such as Mn2+, Cu2+, and Co2+. The most effective metal ion is Mn2+[2]. |
| In Vivo | Rat liver sub-mitochondrial particles also generated hydroxyl radical in the presence of NADH and Rifamycin S. NADH dehydrogenase (complex I) as the major component involved in the reduction of Rifamycin S. Compared to NADPH, NADH is almost as effective (Rifamycin S) in catalyzing the interactions of these antibiotics with rat liver microsomes. Rifamycin S is shown to be readily reduced to Rifamycin SV, the corresponding hydroquinone by Fe(II). Rifamycin S forms a detectable Fe(II)-(Rifamycin S)3 complex. The Fe:ATP induced lipid peroxidation is completely inhibited by Rifamycin S. Rifamycin S can interact with rat liver microsomes to undergo redox-cycling, with the subsequent production of hydroxyl radicals when iron complexes are present[1]. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 917.4±65.0 °C at 760 mmHg |
| Melting Point | 179-181ºC (dec.) |
| Molecular Formula | C37H45NO12 |
| Molecular Weight | 695.753 |
| Flash Point | 508.6±34.3 °C |
| Exact Mass | 695.294189 |
| PSA | 194.99000 |
| LogP | 2.87 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.605 |
| InChIKey | BTVYFIMKUHNOBZ-PZCBORFTSA-N |
| SMILES | COC1C=COC2(C)Oc3c(C)c(O)c4c(c3C2=O)C(=O)C=C(NC(=O)C(C)=CC=CC(C)C(O)C(C)C(O)C(C)C(OC(C)=O)C1C)C4=O |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RTECS | KD1925000 |
|---|---|
| HS Code | 2941903000 |
|
~92% 
RifamycinS CAS#:13553-79-2 |
| Literature: Seong, Baik Lin; Han, Moon Hi Chemistry Letters, 1982 , p. 627 - 628 |
|
~% 
RifamycinS CAS#:13553-79-2 |
| Literature: Journal of the Chemical Society, Chemical Communications, , p. 395 - 396 |
|
~% 
RifamycinS CAS#:13553-79-2 |
| Literature: Journal of the Chemical Society, Chemical Communications, , p. 395 - 396 |
| HS Code | 2941903000 |
|---|
|
Polyketide construction via hydrohydroxyalkylation and related alcohol C-H functionalizations: reinventing the chemistry of carbonyl addition.
Nat. Prod. Rep. 31(4) , 504-13, (2014) Despite the longstanding importance of polyketide natural products in human medicine, nearly all commercial polyketide-based drugs are prepared through fermentation or semi-synthesis. The paucity of m... |
|
|
Synthesis and structure-activity relationships of novel substituted 8-amino, 8-thio, and 1,8-pyrazole congeners of antitubercular rifamycin S and rifampin.
Bioorg. Med. Chem. Lett. 21 , 6094-9, (2011) A series of rifamycin S and rifampin analogues incorporating substituted 8-amino, 8-thio, and 1,8-pyrazole substituents has been synthesized. The compounds were made by activation of the C-8 phenol as... |
|
|
Direct generation of acyclic polypropionate stereopolyads via double diastereo- and enantioselective iridium-catalyzed crotylation of 1,3-diols: beyond stepwise carbonyl addition in polyketide construction.
J. Am. Chem. Soc. 133(32) , 12795-800, (2011) Under the conditions of transfer hydrogenation employing the cyclometalated iridium catalyst (R)-I derived from [Ir(cod)Cl](2), allyl acetate, 4-cyano-3-nitrobenzoic acid, and the chiral phosphine lig... |
|
Name: Antibacterial activity against Staphylococcus aureus 2999
Source: ChEMBL
Target: Staphylococcus aureus
External Id: CHEMBL998299
|
|
Name: Antibacterial activity against Escherichia coli 1074
Source: ChEMBL
Target: Escherichia coli
External Id: CHEMBL998304
|
|
Name: Antibacterial activity against Haemophilus influenzae
Source: ChEMBL
Target: Haemophilus influenzae
External Id: CHEMBL998302
|
|
Name: Antibacterial activity against Neisseria gonorrhoeae
Source: ChEMBL
Target: Neisseria gonorrhoeae
External Id: CHEMBL998303
|
|
Name: Antibacterial activity against Streptococcus pneumoniae
Source: ChEMBL
Target: Streptococcus pneumoniae
External Id: CHEMBL998300
|
|
Name: Antibacterial activity against Streptococcus faecalis 1362/3
Source: ChEMBL
Target: Enterococcus faecalis
External Id: CHEMBL998301
|
|
Name: Antimicrobial activity against Escherichia coli EC2880 mutant after 16 to 20 hrs
Source: ChEMBL
Target: Escherichia coli
External Id: CHEMBL1914230
|
|
Name: Antimicrobial activity against wild type Escherichia coli DH5[alpha] after 16 to 20 h...
Source: ChEMBL
Target: Escherichia coli
External Id: CHEMBL1914229
|
|
Name: Antibacterial activity against rifamycin-sensitive Staphylococcus aureus CB190 ATCC 2...
Source: ChEMBL
Target: Staphylococcus aureus
External Id: CHEMBL899818
|
| Rifamycin,1,4-dideoxy-1,4-dihydro-1,4-dioxo |
| UNII-PI53N820JV |
| rifomycin-S |
| rifamycin-S |
| (7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-2,15,17-Trihydroxy-11-methoxy-3,7,12,14,16,18,22-heptamethyl-6,23,27,29-tetraoxo-8,30-dioxa-24-azatetracyclo[23.3.1.1.0]triaconta-1(28),2,4,9,19,21,25-heptaen-13-yl acetate |
| NCI 144-130 |
| (2S,12Z,14E,16S,17S,18R,19R,20R,21S,22R,23S,24E)-5,17,19-trihydroxy-23-methoxy-2,4,12,16,18,20,22-heptamethyl-1,6,9,11-tetraoxo-1,2,6,9-tetrahydro-2,7-(epoxypentadeca[1,11,13]trienoimino)naphtho[2,1-b]furan-21-yl acetate |
| EINECS 236-938-4 |
| (12S,3E,5S,13E,15Z)-7t-acetoxy-15,9c,11t-trihydroxy-5r-methoxy-12,4,6t,8c,10c,12t,16-heptamethyl-2-oxa-18-aza-1(2,7)-naphtho[2,1-b]furana-cyclooctadecaphane-3,13,15-triene-11,6,9,17-tetraone |
| rifaximin S |
| O1,O4-didehydro-rifamycin |
| (7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-2,15,17-Trihydroxy-11-methoxy-3,7,12,14,16,18,22-heptamethyl-6,23,27,29-tetraoxo-8,30-dioxa-24-azatetracyclo[23.3.1.1.0]triaconta-1(28),2,4,9 ,19,21,25-heptaen-13-yl acetate |
| 1,4-Dideoxy-1,4-dihydro-1,4-dioxorifamycin |
| RifamycinS |

 CAS#:72428-95-6
CAS#:72428-95-6 CAS#:59232-88-1
CAS#:59232-88-1