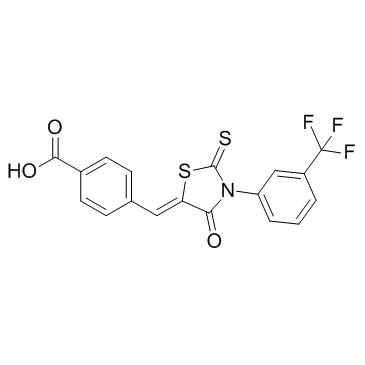
4-Thiazolidinone,2-thioxo-3-[3-(trifluoromethyl)phenyl]
![4-Thiazolidinone,2-thioxo-3-[3-(trifluoromethyl)phenyl] Structure](https://image.chemsrc.com/caspic/455/315-08-2.png)
4-Thiazolidinone,2-thioxo-3-[3-(trifluoromethyl)phenyl] structure
|
Common Name | 4-Thiazolidinone,2-thioxo-3-[3-(trifluoromethyl)phenyl] | ||
|---|---|---|---|---|
| CAS Number | 315-08-2 | Molecular Weight | 277.28600 | |
| Density | 1.58g/cm3 | Boiling Point | 338.1ºC at 760mmHg | |
| Molecular Formula | C10H6F3NOS2 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 158.3ºC | |
| Name | 2-sulfanylidene-3-[3-(trifluoromethyl)phenyl]-1,3-thiazolidin-4-one |
|---|
| Density | 1.58g/cm3 |
|---|---|
| Boiling Point | 338.1ºC at 760mmHg |
| Molecular Formula | C10H6F3NOS2 |
| Molecular Weight | 277.28600 |
| Flash Point | 158.3ºC |
| Exact Mass | 276.98400 |
| PSA | 77.70000 |
| LogP | 3.13510 |
| Index of Refraction | 1.633 |
| InChIKey | MRDJXDJVVCNDND-UHFFFAOYSA-N |
| SMILES | O=C1CSC(=S)N1c1cccc(C(F)(F)F)c1 |
| HS Code | 2934999090 |
|---|
|
~% ![4-Thiazolidinone,2-thioxo-3-[3-(trifluoromethyl)phenyl] Structure](https://image.chemsrc.com/caspic/455/315-08-2.png)
4-Thiazolidinon... CAS#:315-08-2 |
| Literature: US2011/105565 A1, ; Page/Page column 26-28 ; |
|
~52% ![4-Thiazolidinone,2-thioxo-3-[3-(trifluoromethyl)phenyl] Structure](https://image.chemsrc.com/caspic/455/315-08-2.png)
4-Thiazolidinon... CAS#:315-08-2 |
| Literature: Journal of Medicinal Chemistry, , vol. 51, # 24 p. 7843 - 7854 |
|
~% ![4-Thiazolidinone,2-thioxo-3-[3-(trifluoromethyl)phenyl] Structure](https://image.chemsrc.com/caspic/455/315-08-2.png)
4-Thiazolidinon... CAS#:315-08-2 |
| Literature: US2011/105565 A1, ; |
| HS Code | 2934999090 |
|---|---|
| Summary | 2934999090. other heterocyclic compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Name: Discovery of Small Molecules to Inhibit Human Cytomegalovirus Nuclear Egress
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: HCMV UL50
External Id: HMS1262
|
|
Name: Inhibition of HIV1 3B gp41 six-helix bundle formation after 30 mins by native-polyacr...
Source: ChEMBL
Target: N/A
External Id: CHEMBL1017070
|
|
Name: Cytotoxicity against human MT2 cells after 4 days by XTT assay
Source: ChEMBL
Target: MT2
External Id: CHEMBL1017068
|
|
Name: Selectivity index, ratio of CC50 for human MT2 cells to EC50 for HIV1 3B
Source: ChEMBL
Target: N/A
External Id: CHEMBL1017069
|
|
Name: Antiviral activity against HIV1 3B in human MT2 cells assessed as inhibition of p24 a...
Source: ChEMBL
Target: Human immunodeficiency virus 1
External Id: CHEMBL1017067
|
|
Name: Cell-based high throughput primary assay to identify activators of GPR151
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=G-protein coupled receptor 151; AltName: Full=G-protein coupled receptor PGR7; AltName: Full=GPCR-2037; AltName: Full=Galanin receptor 4; AltName: Full=Galanin-receptor-like protein; Short=GalRL
External Id: GPR151_PHUNTER_AG_LUMI_1536_1X%ACT
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify activators of...
Source: The Scripps Research Institute Molecular Screening Center
Target: N/A
External Id: FBW7_ACT_ALPHA_1536_1X%ACT PRUN
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify inhibitors of...
Source: The Scripps Research Institute Molecular Screening Center
External Id: MITF_INH_Alpha_1536_1X%INH PRUN
|
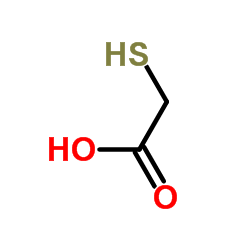
![Acetic acid,2,2'-[carbonothioylbis(thio)]bis structure](https://image.chemsrc.com/caspic/324/6326-83-6.png)
 CAS#:307510-92-5
CAS#:307510-92-5