2-(4-chlorophenyl)-5-methyl-1,3-benzoxazole
Modify Date: 2025-09-21 10:18:38
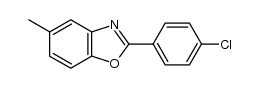
2-(4-chlorophenyl)-5-methyl-1,3-benzoxazole structure
|
Common Name | 2-(4-chlorophenyl)-5-methyl-1,3-benzoxazole | ||
|---|---|---|---|---|
| CAS Number | 35875-75-3 | Molecular Weight | 243.68800 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C14H10ClNO | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
| Name | 2-(4-chlorophenyl)-5-methyl-1,3-benzoxazole |
|---|---|
| Synonym | More Synonyms |
| Molecular Formula | C14H10ClNO |
|---|---|
| Molecular Weight | 243.68800 |
| Exact Mass | 243.04500 |
| PSA | 26.03000 |
| LogP | 4.45660 |
| InChIKey | GSCLPYWGXHGCOK-UHFFFAOYSA-N |
| SMILES | Cc1ccc2oc(-c3ccc(Cl)cc3)nc2c1 |
|
Name: Discovery of Small Molecules to Inhibit Human Cytomegalovirus Nuclear Egress
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: HCMV UL50
External Id: HMS1262
|
|
Name: qHTS assay for inhibitors of human lactate dehydrogenase
Source: NCGC
Target: N/A
External Id: LDHA
|
|
Name: Screen for inhibitors of RMI FANCM (MM2) intereaction
Source: 11908
Target: N/A
External Id: RMI-FANCM-MM2
|
|
Name: Primary qHTS Assay for Foot and Mouth Disease Virus Antivirals against NCGC Sytravon ...
Source: NCGC
External Id: stopgo-p2-SytraCBC-dual-FF
|
|
Name: High-throughput screen for inhibitors of the GIV GBA-motif interaction with Galpha-i ...
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS1303
|
|
Name: Antimicrobial activity against Bacillus subtilis ATCC 6633 after 24 hrs
Source: ChEMBL
Target: Bacillus subtilis
External Id: CHEMBL974326
|
|
Name: A screen for compounds that inhibit the activity of LtaS in Staphylococcus aureus
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS979
|
|
Name: Primary qHTS Assay for Foot and Mouth Disease Virus Antivirals against NCGC Sytravon ...
Source: NCGC
External Id: stopgo-p2-SytraCBC-dual-Ren
|
Total 8, Current Page 1 of 1
1
| 2-(4-chlorophenyl)-5-methylbenzo[d]oxazole |
| 2-(4-chloro-phenyl)-5-methyl-benzooxazole |
| 2-(4-CHLORO-PHENYL)-5-METHYL-BENZOOXAZOLE |
| 2-(4-chlorophenyl)-5-methylbenzoxazole |
| 2-(4-chloro-phenyl)-5-methylbenzoxazole |