Oleandomycin
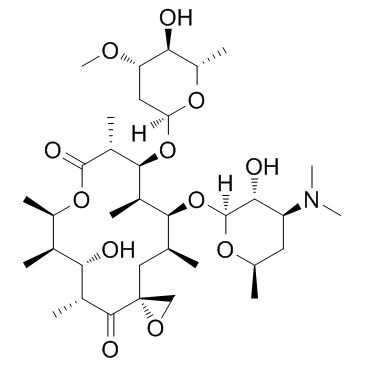
Oleandomycin structure
|
Common Name | Oleandomycin | ||
|---|---|---|---|---|
| CAS Number | 3922-90-5 | Molecular Weight | 687.858 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 802.6±65.0 °C at 760 mmHg | |
| Molecular Formula | C35H61NO12 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 439.2±34.3 °C | |
Use of OleandomycinOleandomycin is a macrolide antibiotic structurally closely related to Erythromycin. Oleandomycin is similar to Erythromycin with antimicrobial activity. |
| Name | oleandomycin |
|---|---|
| Synonym | More Synonyms |
| Description | Oleandomycin is a macrolide antibiotic structurally closely related to Erythromycin. Oleandomycin is similar to Erythromycin with antimicrobial activity. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 802.6±65.0 °C at 760 mmHg |
| Molecular Formula | C35H61NO12 |
| Molecular Weight | 687.858 |
| Flash Point | 439.2±34.3 °C |
| Exact Mass | 687.419373 |
| PSA | 165.98000 |
| LogP | 1.23 |
| Vapour Pressure | 0.0±6.4 mmHg at 25°C |
| Index of Refraction | 1.533 |
| InChIKey | RZPAKFUAFGMUPI-DDSISPHDSA-N |
| SMILES | COC1CC(OC2C(C)C(=O)OC(C)C(C)C(O)C(C)C(=O)C3(CO3)CC(C)C(OC3OC(C)CC(N(C)C)C3O)C2C)OC(C)C1O |
| Storage condition | 2-8℃ |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
|
~% 
Oleandomycin CAS#:3922-90-5 |
| Literature: Tatsuta; Kobayashi; Gunji; Masuda Tetrahedron Letters, 1988 , vol. 29, # 32 p. 3975 - 3978 |
|
~% 
Oleandomycin CAS#:3922-90-5 |
| Literature: Tatsuta; Kobayashi; Gunji; Masuda Tetrahedron Letters, 1988 , vol. 29, # 32 p. 3975 - 3978 |
|
~% 
Oleandomycin CAS#:3922-90-5 |
| Literature: Tatsuta; Kobayashi; Gunji; Masuda Tetrahedron Letters, 1988 , vol. 29, # 32 p. 3975 - 3978 |
|
~% 
Oleandomycin CAS#:3922-90-5 |
| Literature: Tatsuta; Kobayashi; Gunji; Masuda Tetrahedron Letters, 1988 , vol. 29, # 32 p. 3975 - 3978 |
|
~% 
Oleandomycin CAS#:3922-90-5 |
| Literature: Tatsuta; Kobayashi; Gunji; Masuda Tetrahedron Letters, 1988 , vol. 29, # 32 p. 3975 - 3978 |
|
~% 
Oleandomycin CAS#:3922-90-5 |
| Literature: Tatsuta; Kobayashi; Gunji; Masuda Tetrahedron Letters, 1988 , vol. 29, # 32 p. 3975 - 3978 |
|
~% 
Oleandomycin CAS#:3922-90-5 |
| Literature: Tatsuta; Kobayashi; Gunji; Masuda Tetrahedron Letters, 1988 , vol. 29, # 32 p. 3975 - 3978 |
| Precursor 8 | |
|---|---|
| DownStream 3 | |
|
Name: pKa (acid-base dissociation constant) as determined by Avdeef ref: DOI: 10.1002/04714...
Source: ChEMBL
Target: N/A
External Id: CHEMBL2449007
|
|
Name: Minimum inhibitory concentration against Pasteurella multocida.
Source: ChEMBL
Target: Pasteurella multocida
External Id: CHEMBL763912
|
|
Name: Vibrio cholerae assay for pro-quorum sensing small molecules
Source: Southern Research Specialized Biocontainment Screening Center
Target: cyclic AMP receptor protein [Vibrio cholerae O1 biovar El Tor str. N16961]
External Id: Vcholera_QS_Pilot
|
|
Name: Antibacterial activity against Escherichia coli AG102MB harboring pS1328-based acrB g...
Source: ChEMBL
Target: Escherichia coli
External Id: CHEMBL1247932
|
|
Name: Antibacterial activity against Escherichia coli AG102MB harboring pP10-based acrB gen...
Source: ChEMBL
Target: Escherichia coli
External Id: CHEMBL1247931
|
|
Name: Antibacterial activity against Escherichia coli AG102MB harboring naive acrB gene
Source: ChEMBL
Target: Escherichia coli
External Id: CHEMBL1247930
|
|
Name: Antibacterial activity against Escherichia coli AG102MB harboring pSport1-based acrB ...
Source: ChEMBL
Target: Escherichia coli
External Id: CHEMBL1247929
|
|
Name: The negative logarithm of the fraction of singly charged molecules.
Source: ChEMBL
Target: N/A
External Id: CHEMBL633444
|
| (3R,5R,6S,7R,8R,11R,12S,13R,14S,15S)-6-hydroxy-5,7,8,11,13,15-hexamethyl-4,10-dioxo-14-[3,4,6-trideoxy-3-(dimethylamino)-β-D-xylo-hexopyranosyloxy]-1,9-dioxaspiro[2.13]hexadec-12-yl 2,6-dideoxy-3-O-methyl-α-L-arabino-hexopyranoside |
| (3R,5R,6S,7R,8R,11R,12S,13R,14S,15S)-6-Hydroxy-5,7,8,11,13,15-hexamethyl-4,10-dioxo-14-{[3,4,6-trideoxy-3-(dimethylamino)-β-D-xylo-hexopyranosyl]oxy}-1,9-dioxaspiro[2.13]hexadec-12-yl 2,6-dideoxy-3-O-methyl-α-L-arabino-hexopyranoside |
| (3R,5R,6S,7R,8R,11R,12S,13R,14S,15S)-14-{[(2S,3R,4S,6R)-4-(Dimethylamino)-3-hydroxy-6-methyltetrahydro-2H-pyran-2-yl]oxy}-6-hydroxy-12-{[(2R,4S,5S,6S)-5-hydroxy-4-methoxy-6-methyltetrahydro-2H-pyran-2-yl]oxy}-5,7,8,11,13,15-hexamethyl-1,9-dioxaspiro[2.13]hexadecane-4,10-dione |
| (3R,5R,6S,7R,8R,11R,12S,13R,14S,15S)-6-Hydroxy-5,7,8,11,13,15-hexamethyl-4,10-dioxo-14-{[3,4,6-trideoxy-3-(dimethylamino)-β-D-xylo-hexopyranosyl]oxy}-1,9-dioxaspiro[2.13]hexadec-12-yl 2,6-dideoxy-3 -O-methyl-α-L-arabino-hexopyranoside |
| Oleandomycin |
| 1-Propanaminium,N-(carboxymethyl)-N,N-dimethyl-3-[(1-oxo-9-octadecenyl)amino]-,hydroxide,inner salt,(Z) |
| Oleamidopropyl dimethyl glycine |
| (Z)-(Carboxymethyl)dimethyl-3-((1-oxo-9-octadecenyl)amino)propylammonium hydroxide |
| N-Oleoylamidopropyl-N,N-dimethylbetaine |
| Oleamidopropyl betaine |
![(1S,2R,2'R,5R,6R,7R,8R,9R,12S,13S,17R)-15-(4-bromophenyl)-7,9-dihydroxy-2,5,6,8,12,17-hexamethyl-4,14,16-trioxaspiro[bicyclo[11.3.1]heptadecane-10,2'-oxiran]-3-one structure](https://image.chemsrc.com/caspic/175/119870-69-8.png)
![(1S,2R,5R,6R,7R,8R,9R,12S,13S,17R)-15-(4-bromophenyl)-7,9-dihydroxy-2,5,6,8,12,17-hexamethyl-10-methylene-4,14,16-trioxabicyclo[11.3.1]heptadecan-3-one structure](https://image.chemsrc.com/caspic/408/119887-69-3.png)
![(1S,2R,2'R,5R,6R,7S,8R,12S,13S,17R)-15-(4-bromophenyl)-7-hydroxy-2,5,6,8,12,17-hexamethyl-4,14,16-trioxaspiro[bicyclo[11.3.1]heptadecane-10,2'-oxirane]-3,9-dione structure](https://image.chemsrc.com/caspic/310/119870-70-1.png)
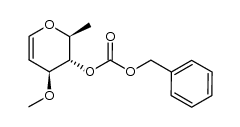
![(2S,3R,4S,6R)-2-(((3R,5R,6S,7R,8R,11R,12S,13S,14S,15S)-6,12-dihydroxy-5,7,8,11,13,15-hexamethyl-4,10-dioxo-1,9-dioxaspiro[2.13]hexadecan-14-yl)oxy)-4-(dimethylamino)-6-methyltetrahydro-2H-pyran-3-yl acetate structure](https://image.chemsrc.com/caspic/196/119870-73-4.png)
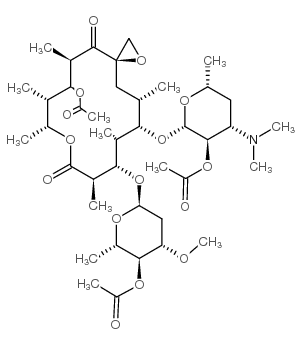 CAS#:2751-09-9
CAS#:2751-09-9![6-[4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy-8,14-dihydroxy-5,7,9,12,13,15-hexamethyl-1,11-dioxaspiro[2.13]hexadecane-10,16-dione structure](https://image.chemsrc.com/caspic/019/4166-78-3.png) CAS#:4166-78-3
CAS#:4166-78-3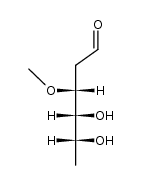 CAS#:13089-77-5
CAS#:13089-77-5