Gabapentin enacarbil
Modify Date: 2025-08-20 08:57:26
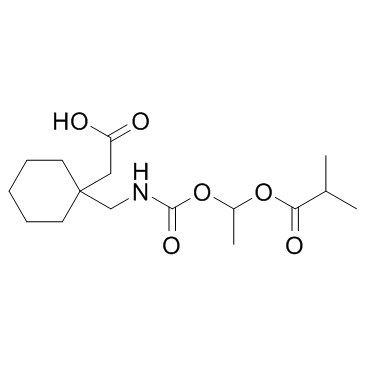
Gabapentin enacarbil structure
|
Common Name | Gabapentin enacarbil | ||
|---|---|---|---|---|
| CAS Number | 478296-72-9 | Molecular Weight | 329.389 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 482.0±20.0 °C at 760 mmHg | |
| Molecular Formula | C16H27NO6 | Melting Point | 65ºC | |
| MSDS | N/A | Flash Point | 245.3±21.8 °C | |
Use of Gabapentin enacarbilGabapentin enacarbil (XP-13512) is a prodrug for the anticonvulsant and analgesic drug gabapentin.IC50 Value: Target: Calcium ChannelGabapentin enacarbil is an actively transported prodrug of gabapentin that provides sustained dose-proportional exposure to gabapentin and predictable bioavailability.in vitro: The prodrug (XP-13512) demonstrated active apical to basolateral transport across Caco-2 cell monolayers and pH-dependent passive permeability across artificial membranes. XP13512 inhibited uptake of (14)C-lactate by human embryonic kidney cells expressing monocarboxylate transporter type-1, and direct uptake of prodrug by these cells was confirmed using liquid chromatography-tandem mass spectrometry. XP13512 inhibited uptake of (3)H-biotin into Chinese hamster ovary cells overexpressing human sodium-dependent multivitamin transporter (SMVT) [1].in vivo: In 4 studies of healthy volunteers (136 subjects total), the pharmacokinetics of XP13512 immediate- and extended-release formulations were compared with those of oral gabapentin. XP13512 immediate-release (up to 2800 mg single dose and 2100 mg twice daily) was well absorbed (>68%, based on urinary recovery of gabapentin), converted rapidly to gabapentin, and provided dose-proportional exposure, whereas absorption of oral gabapentin declined with increasing doses to <27% at 1200 mg. Compared with 600 mg gabapentin, an equimolar XP13512 extended-release dose provided extended gabapentin exposure (time to maximum concentration, 8.4 vs 2.7 hours) and superior bioavailability (74.5% vs 36.6%) [2].Toxicity: Gabapentin's most common side effects in adult patients include dizziness, fatigue, weight gain, drowsiness, and peripheral edema (swelling of extremities). |
| Name | gabapentin enacarbil |
|---|---|
| Synonym | More Synonyms |
| Description | Gabapentin enacarbil (XP-13512) is a prodrug for the anticonvulsant and analgesic drug gabapentin.IC50 Value: Target: Calcium ChannelGabapentin enacarbil is an actively transported prodrug of gabapentin that provides sustained dose-proportional exposure to gabapentin and predictable bioavailability.in vitro: The prodrug (XP-13512) demonstrated active apical to basolateral transport across Caco-2 cell monolayers and pH-dependent passive permeability across artificial membranes. XP13512 inhibited uptake of (14)C-lactate by human embryonic kidney cells expressing monocarboxylate transporter type-1, and direct uptake of prodrug by these cells was confirmed using liquid chromatography-tandem mass spectrometry. XP13512 inhibited uptake of (3)H-biotin into Chinese hamster ovary cells overexpressing human sodium-dependent multivitamin transporter (SMVT) [1].in vivo: In 4 studies of healthy volunteers (136 subjects total), the pharmacokinetics of XP13512 immediate- and extended-release formulations were compared with those of oral gabapentin. XP13512 immediate-release (up to 2800 mg single dose and 2100 mg twice daily) was well absorbed (>68%, based on urinary recovery of gabapentin), converted rapidly to gabapentin, and provided dose-proportional exposure, whereas absorption of oral gabapentin declined with increasing doses to <27% at 1200 mg. Compared with 600 mg gabapentin, an equimolar XP13512 extended-release dose provided extended gabapentin exposure (time to maximum concentration, 8.4 vs 2.7 hours) and superior bioavailability (74.5% vs 36.6%) [2].Toxicity: Gabapentin's most common side effects in adult patients include dizziness, fatigue, weight gain, drowsiness, and peripheral edema (swelling of extremities). |
|---|---|
| Related Catalog | |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 482.0±20.0 °C at 760 mmHg |
| Melting Point | 65ºC |
| Molecular Formula | C16H27NO6 |
| Molecular Weight | 329.389 |
| Flash Point | 245.3±21.8 °C |
| Exact Mass | 329.183838 |
| PSA | 101.93000 |
| LogP | 3.07 |
| Vapour Pressure | 0.0±2.6 mmHg at 25°C |
| Index of Refraction | 1.481 |
| InChIKey | TZDUHAJSIBHXDL-UHFFFAOYSA-N |
| SMILES | CC(OC(=O)NCC1(CC(=O)O)CCCCC1)OC(=O)C(C)C |
| Storage condition | 2-8℃ |
| Precursor 9 | |
|---|---|
| DownStream 0 | |
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of VER...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4513082
|
|
Name: Oral bioavailability in cynomolgus monkey assessed as gabapentin at 36 mg/kg
Source: ChEMBL
Target: Macaca fascicularis
External Id: CHEMBL3118799
|
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of Cac...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303805
|
|
Name: Cytotoxicity counterscreen for inhibitors of SARS-CoV-2 cell entry
Source: NCGC
Target: N/A
External Id: TRND-SARS-CoV-2-cytotox-48hr
|
|
Name: SARS-CoV-2 3CL-Pro protease inhibition percentage at 20µM by FRET kind of response f...
Source: ChEMBL
Target: Replicase polyprotein 1ab
External Id: CHEMBL4495582
|
|
Name: Primary qHTS to identify inhibitors of SARS-CoV-2 cell entry
Source: NCGC
External Id: TRND-SARS-CoV-2-PP
|
|
Name: Antiviral activity against SARS-CoV-2 (USA-WA1/2020 strain) measured by imaging in HR...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303810
|
|
Name: Enzymatic assay of human HDAC6 with commercial peptide substrate
Source: ChEMBL
Target: Histone deacetylase 6
External Id: CHEMBL4808149
|
|
Name: Enzymatic assay of human HDAC6 with custom peptide substrate
Source: ChEMBL
Target: Histone deacetylase 6
External Id: CHEMBL4808150
|
|
Name: Primary qHTS for inhibitors of NSP2Pro chikungunya virus (CHIKV)
Source: NCGC
External Id: APP-Toga-CHIKV-nsp2-p
|
Total 15, Current Page 1 of 2
1
2
| {[(1-isobutanoyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic acid |
| {1-[({[1-(Isobutyryloxy)ethoxy]carbonyl}amino)methyl]cyclohexyl}acetic acid |
| Solzira |
| UNII-75OCL1SPBQ |
| UNII:75OCL1SPBQ |
| Horizant |
| 2-[1-[[1-(2-methylpropanoyloxy)ethoxycarbonylamino]methyl]cyclohexyl]acetic acid |
| ASP-8825 |
| Gabapentin enacarbil |
| Xenoport |
![Propanoic acid, 2-Methyl-, 1-[[[(2,5-dioxo-1-pyrrolidinyl)oxy]carbonyl]oxy]ethyl ester Structure](https://image.chemsrc.com/caspic/232/860035-10-5.png) CAS#:860035-10-5
CAS#:860035-10-5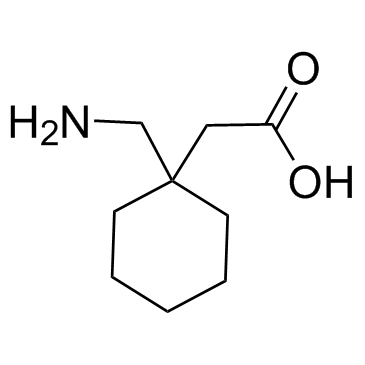 CAS#:60142-96-3
CAS#:60142-96-3![1-[(chlorocarbonyl)oxy]ethyl 2-methylpropionate Structure](https://image.chemsrc.com/caspic/312/1164116-60-2.png) CAS#:1164116-60-2
CAS#:1164116-60-2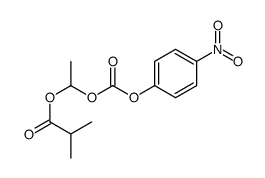 CAS#:194995-47-6
CAS#:194995-47-6![1-{[(α-chloroethoxy)carbonyl]aminomethyl}-1-cyclohexane acetic acid Structure](https://image.chemsrc.com/caspic/356/850479-14-0.png) CAS#:850479-14-0
CAS#:850479-14-0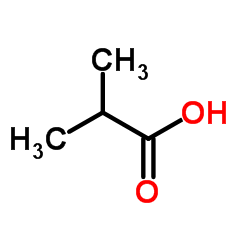 CAS#:79-31-2
CAS#:79-31-2 CAS#:1201785-64-9
CAS#:1201785-64-9 CAS#:1201785-62-7
CAS#:1201785-62-7 CAS#:649748-02-7
CAS#:649748-02-7