Monoctanoin
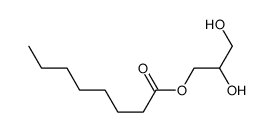
Monoctanoin structure
|
Common Name | Monoctanoin | ||
|---|---|---|---|---|
| CAS Number | 502-54-5 | Molecular Weight | 218.29000 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C11H22O4 | Melting Point | N/A | |
| MSDS | USA | Flash Point | N/A | |
Use of MonoctanoinGlyceryl 1-monooctanoate is a glycerol monolaurate derivative. Glyceryl 1-monooctanoate is a broad-spectrum antimicrobial, suppresses the growth of pathogenic yeast (Candida albicans and Candida parapsilosis), as well as Gram-positive (Staphylococcus aureus) and Gram-negative (Escherichia coli, Klebsiella pneumoniae) bacteria[1]. |
| Name | 1-monooctanoylglycerol |
|---|---|
| Synonym | More Synonyms |
| Description | Glyceryl 1-monooctanoate is a glycerol monolaurate derivative. Glyceryl 1-monooctanoate is a broad-spectrum antimicrobial, suppresses the growth of pathogenic yeast (Candida albicans and Candida parapsilosis), as well as Gram-positive (Staphylococcus aureus) and Gram-negative (Escherichia coli, Klebsiella pneumoniae) bacteria[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Glyceryl 1-monooctanoate (0-32.5 mg/L; 30 d) has antifungal activity against saprophytic fungi. Glyceryl 1-monooctanoate inhibits the growth of Aspergillus niger, Penicillium roqueforti, Penicillium jensenii, Alternaria sp., Phoma sp[1]. Glyceryl 1-monooctanoate (0-32.5 mg/L; 30 d) has antifungal activity against pathogenic fungi. Glyceryl 1-monooctanoate inhibits the growth of Trichophyton mentagrophytes and Trichophyton rubrum[1]. Glyceryl 1-monooctanoate (30 d) has antifungal activity against pathogenic yeast with minimum inhibitory concentration (MIC) of 200 mg/L for Candida albicans[1]. Glyceryl 1-monooctanoate (30 d) has antifungal activity against Gram-positive (Staphylococcus aureus) and Gram-negative (Escherichia coli, Klebsiella pneumoniae) bacteria[1]. |
| References |
| Molecular Formula | C11H22O4 |
|---|---|
| Molecular Weight | 218.29000 |
| Exact Mass | 218.15200 |
| PSA | 66.76000 |
| LogP | 1.24330 |
| InChIKey | GHBFNMLVSPCDGN-UHFFFAOYSA-N |
| SMILES | CCCCCCCC(=O)OCC(O)CO |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
|
How relevant are assembled equilibrium samples in understanding structure formation during lipid digestion?
Eur. J. Pharm. Biopharm. 96 , 117-24, (2015) Lipid-based formulations are gaining interest for use as drug delivery systems for poorly water-soluble drug compounds. During digestion, the lipolysis products self-assemble with endogenous surfactan... |
|
|
Transcutaneous delivery of leflunomide nanoemulgel: Mechanistic investigation into physicomechanical characteristics, in vitro anti-psoriatic and anti-melanoma activity.
Int. J. Pharm. 487 , 148-56, (2015) The present study is a mechanistic validation of 'proof of concept' of effective topical delivery of leflunomide (LFD) nanoemulgel for localized efficient treatment of psoriatic lesions as well as mel... |
|
|
Solid self-nanoemulsifying drug delivery system (S-SNEDDS) of darunavir for improved dissolution and oral bioavailability: In vitro and in vivo evaluation.
Eur. J. Pharm. Sci. 74 , 1-10, (2015) The current study was aimed to investigate the potential of solid self-nanoemulsifying drug delivery system (S-SNEDDS) composed of Capmul MCM C8 (oil), Tween 80 (surfactant) and Transcutol P (co-surfa... |
| 1-Octanoyl-rac-glycerol |
| MONOCAPRYLIN |
| 1-Capryloyl-rac-glycerol Monocaprylin |