Tanshinone IIA
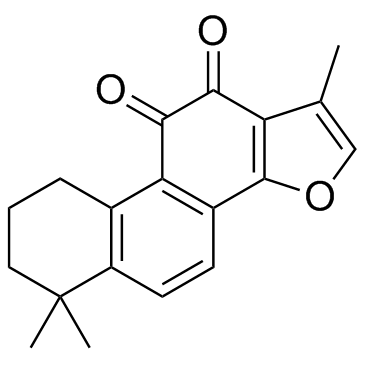
Tanshinone IIA structure
|
Common Name | Tanshinone IIA | ||
|---|---|---|---|---|
| CAS Number | 568-72-9 | Molecular Weight | 294.344 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 480.7±44.0 °C at 760 mmHg | |
| Molecular Formula | C19H18O3 | Melting Point | 205-207ºC | |
| MSDS | Chinese USA | Flash Point | 236.4±21.1 °C | |
Use of Tanshinone IIATanshinone IIA (Tan IIA) is one of the main fat-soluble compositions in the root of red-rooted salvia. Tanshinone IIA may suppress angiogenesis by targeting the protein kinase domains of VEGF/VEGFR2. |
| Name | Tanshinone IIA |
|---|---|
| Synonym | More Synonyms |
| Description | Tanshinone IIA (Tan IIA) is one of the main fat-soluble compositions in the root of red-rooted salvia. Tanshinone IIA may suppress angiogenesis by targeting the protein kinase domains of VEGF/VEGFR2. |
|---|---|
| Related Catalog | |
| Target |
VEGF/VEGFR2[1] |
| In Vitro | The anti-tumor effect of Tanshinone IIA includes inhibiting tumor cell proliferation, disturbing tumor cell cycle, promoting tumor cell apoptosis, and inhibiting tumor cell invasion and transfer. Tanshinone IIA has anti-proliferative effects on A549 cells: the IC50 of Tanshinone IIA after 24, 48 and 72 h are 145.3, 30.95 and 11.49 μM, respectively. The CCK-8 assay is used to evaluate the proliferative activity of A549 cells treated with Tanshinone IIA (2.5-80 μM) for 24, 48 and 72 h, respectively. The CCK-8 results show that Tanshinone IIA can significantly inhibit A549 cell proliferation in a dose- and time-dependent manner. Obvious apoptosis and cell growth inhibition of A549 cells are observed after drug treatment for 48 h (concentrations used are approximately IC50 values: Tanshinone IIA 31 μM on A549). Western blotting finds that 48 h exposures to Tanshinone IIA (31 μM) in A549 cells, downregulates expression of VEGF and VEGFR2 protein in both drug treatment groups vs. vehicle[1]. Tanshinone IIA, one of the most abundant constituents of the root of Salvia miltiorrhiza, protects rat myocardium-derived H9C2 cells against apoptosis. Treatment of H9C2 cells with Tanshinone IIA inhibits angiotensin II-induced apoptosis by downregulating the expression of PTEN (phosphatase and tensin homolog), a tumor suppressor that plays a critical role in apoptosis. Tanshinone IIA inhibits angiotensin II (AngII)-induced apoptosis by downregulating the expression of phosphatase and tensin homolog (PTEN)[2]. Tanshinone IIA decreases the protein expression of EGFR, and IGFR blocking the PI3K/Akt/mTOR pathway in gastric carcinoma AGS cells[3]. |
| Cell Assay | A549 cells are counted in logarithmic phase and 6000 cells (90 μL volume) are placed in 96-well plates. 10 μL varying concentrations of Tanshinone IIA (final concentrations 80, 60, 40, 30, 20, 15, 10, 5 and 2.5 μM) and ADM (final concentrations 8, 4, 2, 1, 0.5 and 0.25 μM) are added into drug groups, while negative control group (vehicle group) is only added 10 μL DMSO or normal saline without Tanshinone IIA or ADM. Cells are incubated for an additional 2 h with CCK-8 reagent (100 μL/mL medium) and the absorbance is read at 450 nm using a microplate reader. Cell proliferation inhibition rates are calculated according to the following formula: the proliferation inhibition ratio (%)=1-[(A1-A4)/(A2-A3)]×100, where, A1 is the OD value of drug experimental group, A2 is the OD value of blank control group, A3 is the OD value of the RPMI1640 medium without cells, and A4 is the OD value of drugs with the same concentration as A1 but without cells. The IC50 value, which represents the concentration of the drug that demonstrates 50% of cell growth inhibition, is calculated by nonlinear regression analysis using GraphPad Prism software[1]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 480.7±44.0 °C at 760 mmHg |
| Melting Point | 205-207ºC |
| Molecular Formula | C19H18O3 |
| Molecular Weight | 294.344 |
| Flash Point | 236.4±21.1 °C |
| Exact Mass | 294.125580 |
| PSA | 47.28000 |
| LogP | 5.47 |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C |
| Index of Refraction | 1.588 |
| InChIKey | HYXITZLLTYIPOF-UHFFFAOYSA-N |
| SMILES | Cc1coc2c1C(=O)C(=O)c1c-2ccc2c1CCCC2(C)C |
| Storage condition | 2-8°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
|
The CO donor CORM-2 inhibits LPS-induced vascular cell adhesion molecule-1 expression and leukocyte adhesion in human rheumatoid synovial fibroblasts.
Br. J. Pharmacol. 171(12) , 2993-3009, (2014) Infection with Gram-negative bacteria has been recognized as an initiator of rheumatoid arthritis, which is characterized by chronic inflammation and infiltration of immune cells. Carbon monoxide (CO)... |
|
|
Modulation of esterified drug metabolism by tanshinones from Salvia miltiorrhiza ("Danshen").
J. Nat. Prod. 76(1) , 36-44, (2013) The roots of Salvia miltiorrhiza ("Danshen") are used in traditional Chinese medicine for the treatment of numerous ailments including cardiovascular disease, hypertension, and ischemic stroke. Extrac... |
|
|
Surfactant-coated graphitized multiwalled carbon nanotubes as the pseudostationary phase in electrokinetic chromatography for the analysis of phytochemical compounds in biological fluids.
Electrophoresis 36(7-8) , 1055-63, (2015) This report describes the use of surfactant-coated graphitized multiwalled carbon nanotubes (SC-GMWNTs) as a novel pseudostationary phase in CE with diode array detection for the determination of phen... |
|
Name: Inhibition of human recombinant telomerase activity in rabbit reticulocytes pre-incub...
Source: ChEMBL
Target: Telomerase reverse transcriptase
External Id: CHEMBL2160442
|
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Induction of ROS generation assessed as hydrogen peroxide production in PBS buffer at...
Source: ChEMBL
Target: N/A
External Id: CHEMBL2160440
|
|
Name: Inhibition of pre-assembled human recombinant telomerase activity in rabbit reticuloc...
Source: ChEMBL
Target: Telomerase reverse transcriptase
External Id: CHEMBL2160441
|
|
Name: Induction of ROS generation assessed as hydrogen peroxide production in PBS buffer at...
Source: ChEMBL
Target: N/A
External Id: CHEMBL2160438
|
|
Name: Induction of ROS generation assessed as hydrogen peroxide production in PBS buffer at...
Source: ChEMBL
Target: N/A
External Id: CHEMBL2160439
|
|
Name: Quantitative High-Throughput drug screen in 47 multiple myeloma cell lines against th...
Source: NCGC
Target: N/A
External Id: s-my-keats_OPM1-m4-1
|
|
Name: Quantitative High-Throughput drug screen in 47 multiple myeloma cell lines against th...
Source: NCGC
Target: N/A
External Id: s-my-OC1MY5-m4-1
|
|
Name: Increase the activity of the Burkholderia fixLJ 2-component system
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: Burkholderia multivorans
External Id: HMS1625
|
| Tanshinone IIA |
| tanshinone II-A |
| Dan Shen Ketone |
| Tanshionesiia |
| Tanshine II |
| TANSHION P.E |
| 1,6,6-Trimethyl-6,7,8,9-tetrahydrophenanthro[1,2-b]furan-10,11-dione |
| SweetOrange |
| MFCD00238692 |
| QS-D-77-4-2 |
| TANSHINONE A |
| tanshiones |
| Tanshinone II |
| TANSHINONES IIA |
 CAS#:35825-57-1
CAS#:35825-57-1 CAS#:2270-59-9
CAS#:2270-59-9 CAS#:578-57-4
CAS#:578-57-4 CAS#:171979-69-4
CAS#:171979-69-4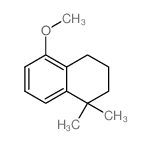 CAS#:33214-70-9
CAS#:33214-70-9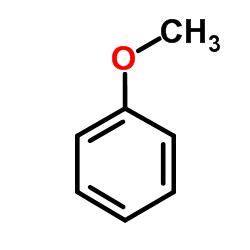 CAS#:100-66-3
CAS#:100-66-3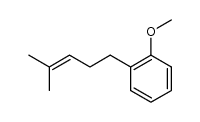 CAS#:171979-70-7
CAS#:171979-70-7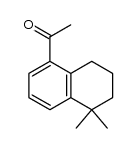 CAS#:171979-68-3
CAS#:171979-68-3
