methyl(2,3,3-trimethyltrinorbornan-2-yl)amine
Modify Date: 2025-08-23 11:22:41
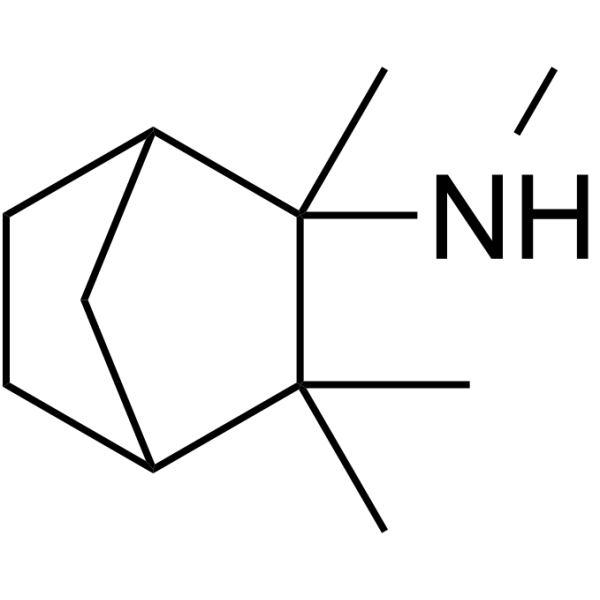
methyl(2,3,3-trimethyltrinorbornan-2-yl)amine structure
|
Common Name | methyl(2,3,3-trimethyltrinorbornan-2-yl)amine | ||
|---|---|---|---|---|
| CAS Number | 60-40-2 | Molecular Weight | 167.29100 | |
| Density | 0.91 | Boiling Point | 189.3ºC at 760 mmHg | |
| Molecular Formula | C11H21N | Melting Point | 245-256ºC | |
| MSDS | N/A | Flash Point | 58.1ºC | |
Use of methyl(2,3,3-trimethyltrinorbornan-2-yl)amineMecamylamine is an orally active, nonselective, noncompetitive nAChR antagonist. Mecamylamine is also a ganglionic blocker. Mecamylamine can across the blood-brain barrier. Mecamylamine can be used in the research of neuropsychiatric disorders, hypertension, antidepressant area[1][2][5]. |
| Name | N,2,2,3-tetramethylbicyclo[2.2.1]heptan-3-amine |
|---|---|
| Synonym | More Synonyms |
| Description | Mecamylamine is an orally active, nonselective, noncompetitive nAChR antagonist. Mecamylamine is also a ganglionic blocker. Mecamylamine can across the blood-brain barrier. Mecamylamine can be used in the research of neuropsychiatric disorders, hypertension, antidepressant area[1][2][5]. |
|---|---|
| Related Catalog | |
| Target |
nAChR[1], histamine receptor[2] |
| In Vitro | Mecamylamine (0.5-9 μM, bath administered) increases the firing frequency of identified 5-HT DRN neurons[1]. Mecamylamine (0.5-9 μM, bath administered) increases the glutamatergic and decreases the GABAergic input of 5-HT DRN neurons[1]. Mecamylamine (1 mM, 5 min) blocks the histamine receptor and the histamine-induced contractions in helically cut strips of rabbit aorta[2]. Mecamylamine (10 μM,48 h) attenuates the effect of nicotine’s action of neuroprotection[3]. Mecamylamine (1-100 nM, 30 min) dose-dependently attenuates endothelial tube formation in HDMVECs[4]. Western Blot Analysis[3] Cell Line: [3] Concentration: 10 μM Incubation Time: 48 h Result: Reduced the nicotine-facilitated increase in ERK1/2. |
| In Vivo | Mecamylamine (subcutaneous pumps, 50 mg/kg/day, 2 days) inhibits Choroidal neovascularization (CNV) in CNV mice model[4]. Mecamylamine (intraperitoneal injection, 0.5-1 mg/kg) has antidepressant-like effects in both the TST (tail suspension test) and FST (forced swim test) in C57BL/6J mice, which are dependent on bothβ2 andα7 subunits[5]. Animal Model: Choroidal neovascularization (CNV) mice model[1] Dosage: 50 mg/kg/day, 2 days Administration: Subcutaneous pumps implanted beneath the skin of the back), 200 μL and mean pumping rate of 0.5 μL/h. Result: Suppressed the development of CNV at Bruch’s membrane rupture sites in the absence of nicotine. Animal Model: C57BL/6J mice[5] Dosage: 0.5-1 mg/kg Administration: Intraperitoneal injection Result: Had no effect in β2 knockout miceand α7 knockout mice, but decreased immobility time in wildtype littermates in the FST. |
| References |
| Density | 0.91 |
|---|---|
| Boiling Point | 189.3ºC at 760 mmHg |
| Melting Point | 245-256ºC |
| Molecular Formula | C11H21N |
| Molecular Weight | 167.29100 |
| Flash Point | 58.1ºC |
| Exact Mass | 167.16700 |
| PSA | 12.03000 |
| LogP | 2.81150 |
| Index of Refraction | 1.4875 |
| InChIKey | IMYZQPCYWPFTAG-UHFFFAOYSA-N |
| SMILES | CNC1(C)C2CCC(C2)C1(C)C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
|
Name: Human nicotinic acetylcholine receptor alpha7 subunit (Nicotinic acetylcholine recept...
Source: IUPHAR-DB
Target: nicotinic acetylcholine receptor alpha7 subunit (Nicotinic acetylcholine receptors) [Homo sapiens]
External Id: 468_Human
|
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: Human nicotinic acetylcholine receptor alpha3 subunit (Nicotinic acetylcholine recept...
Source: IUPHAR-DB
Target: nicotinic acetylcholine receptor alpha3 subunit (Nicotinic acetylcholine receptors) [Homo sapiens]
External Id: 464_Human
|
|
Name: Human nicotinic acetylcholine receptor alpha6 subunit (Nicotinic acetylcholine recept...
Source: IUPHAR-DB
Target: nicotinic acetylcholine receptor alpha6 subunit (Nicotinic acetylcholine receptors) [Homo sapiens]
External Id: 467_Human
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: Human nicotinic acetylcholine receptor alpha2 subunit (Nicotinic acetylcholine recept...
Source: IUPHAR-DB
Target: nicotinic acetylcholine receptor alpha2 subunit (Nicotinic acetylcholine receptors) [Homo sapiens]
External Id: 463_Human
|
|
Name: Primary qHTS for inhibitors of NSP2Pro chikungunya virus (CHIKV)
Source: NCGC
External Id: APP-Toga-CHIKV-nsp2-p
|
|
Name: qHTS Assay for Identifying Compounds that block Entry of Ebola Virus, Screen 2 green ...
Source: NCGC
Target: N/A
External Id: Ebola screen2_EMI141217_green
|
|
Name: qHTS Assay for Identifying Compounds that block Entry of Ebola Virus, Screen 1 blue c...
Source: NCGC
Target: N/A
External Id: Ebola screen1_EMI141217blue
|
Total 431, Current Page 1 of 44
1
2
3
4
5
| EINECS 200-476-1 |