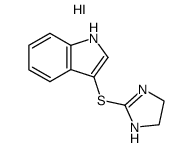
Tinazoline
Modify Date: 2025-11-11 20:07:29
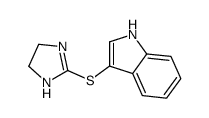
Tinazoline structure
|
Common Name | Tinazoline | ||
|---|---|---|---|---|
| CAS Number | 62882-99-9 | Molecular Weight | 217.29000 | |
| Density | 1.42g/cm3 | Boiling Point | 437.6ºC at 760 mmHg | |
| Molecular Formula | C11H11N3S | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 218.4ºC | |
Use of TinazolineTinazoline, imidazole derivative, is a vasoconstrictor. Tinazoline can as nasal decongestant for the research of common cold[1]. |
| Name | 3-(4,5-Dihydro-1H-imidazol-2-ylsulfanyl)-1H-indole |
|---|---|
| Synonym | More Synonyms |
| Description | Tinazoline, imidazole derivative, is a vasoconstrictor. Tinazoline can as nasal decongestant for the research of common cold[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.42g/cm3 |
|---|---|
| Boiling Point | 437.6ºC at 760 mmHg |
| Molecular Formula | C11H11N3S |
| Molecular Weight | 217.29000 |
| Flash Point | 218.4ºC |
| Exact Mass | 217.06700 |
| PSA | 65.48000 |
| LogP | 1.98360 |
| Index of Refraction | 1.755 |
| InChIKey | JENBDJGHNIOHAJ-UHFFFAOYSA-N |
| SMILES | c1ccc2c(SC3=NCCN3)c[nH]c2c1 |
|
~% 
Tinazoline CAS#:62882-99-9 |
| Literature: Nagarajan, K.; Arya, V. P.; Parthasarathy, T. N.; Shenoy, S. J.; Shah, R. K.; Kulkarni, Y. S. Indian Journal of Chemistry, Section B: Organic Chemistry Including Medicinal Chemistry, 1981 , vol. 20, # 8 p. 672 - 679 |
|
~% 
Tinazoline CAS#:62882-99-9 |
| Literature: Nagarajan, K.; Arya, V. P.; Parthasarathy, T. N.; Shenoy, S. J.; Shah, R. K.; Kulkarni, Y. S. Indian Journal of Chemistry, Section B: Organic Chemistry Including Medicinal Chemistry, 1981 , vol. 20, # 8 p. 672 - 679 |
Total 30, Current Page 1 of 3
1
2
3
| 3-(4,5-dihydro-1H-imidazol-2-ylmethyl)-benzo[d]isoxazole,monohydrochloride |
| 3-(2-Imidazolin-2-yl)methyl-1,2-benzisoxazole hydrochloride |
| 3-(2-imidazolin-2-ylthio)-indole |
| 3-(2-Imidazolin-2-ylthio)-indol |
| 3-(4,5-dihydro-1H-imidazol-2-ylsulfanyl)-indole |