Daurichromenic acid
Modify Date: 2025-08-25 13:51:04
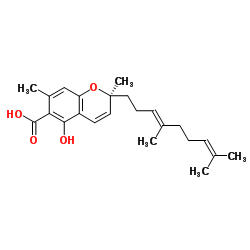
Daurichromenic acid structure
|
Common Name | Daurichromenic acid | ||
|---|---|---|---|---|
| CAS Number | 82003-90-5 | Molecular Weight | 370.48 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 503.7±50.0 °C at 760 mmHg | |
| Molecular Formula | C23H30O4 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 167.1±23.6 °C | |
Use of Daurichromenic acidDaurichromenic acid is a chromene, which can be isolated from the leaves and twigs of Rhododendron dauricum. Daurichromenic acid has potent anti-HIV activity with an EC50 value of 0.00567 μg/mL[1]. |
| Name | (2S)-2-[(3E)-4,8-Dimethyl-3,7-nonadien-1-yl]-5-hydroxy-2,7-dimeth yl-2H-chromene-6-carboxylic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Daurichromenic acid is a chromene, which can be isolated from the leaves and twigs of Rhododendron dauricum. Daurichromenic acid has potent anti-HIV activity with an EC50 value of 0.00567 μg/mL[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 503.7±50.0 °C at 760 mmHg |
| Molecular Formula | C23H30O4 |
| Molecular Weight | 370.48 |
| Flash Point | 167.1±23.6 °C |
| Exact Mass | 370.214417 |
| PSA | 66.76000 |
| LogP | 9.20 |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.554 |
| InChIKey | UYLFTJMQPWWDCW-QHCPKHFHSA-N |
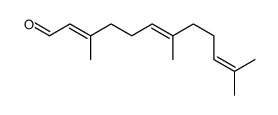
| SMILES | CC(C)=CCCC(C)=CCCC1(C)C=Cc2c(cc(C)c(C(=O)O)c2O)O1 |
|
~58% 
Daurichromenic acid CAS#:82003-90-5 |
| Literature: Liu, Kegang; Woggon, Woif-D. European Journal of Organic Chemistry, 2010 , # 6 p. 1033 - 1036 |
|
~% 
Daurichromenic acid CAS#:82003-90-5 |
| Literature: UNIVERSITY OF IOWA RESEARCH FOUNDATION; JIN, Zhendong; KANG, Ying Patent: WO2004/58738 A1, 2004 ; Location in patent: Page 2/2 ; |
| Precursor 3 | |
|---|---|
| DownStream 0 | |
|
Name: Inhibition of human amyloid beta (1 to 40) aggregation after 24 hrs by ThT fluorescen...
Source: ChEMBL
Target: Amyloid-beta precursor protein
External Id: CHEMBL4603707
|
|
Name: Cytotoxicity against human H9 cells by Coulter counter method
Source: ChEMBL
Target: H9
External Id: CHEMBL5253500
|
|
Name: Antiviral activity against HIV-1 3B in human H9 cells incubated for 4 days by ELISA
Source: ChEMBL
Target: Human immunodeficiency virus 1
External Id: CHEMBL5253499
|
|
Name: Therapeutic index, ratio of IC50 for human H9 cells to EC50 for HIV-1 3B in human H9 ...
Source: ChEMBL
Target: N/A
External Id: CHEMBL5253501
|
|
Name: Therapeutic index, ratio of IC50 for human H9 cells to EC50 for HIV 3B
Source: ChEMBL
Target: N/A
External Id: CHEMBL1176168
|
|
Name: Antiviral activity against HIV1 3B infected in H9 cells assessed as inhibition of vir...
Source: ChEMBL
Target: Human immunodeficiency virus 1
External Id: CHEMBL1176167
|
Total 7, Current Page 1 of 1
1
| (2S)-2-[(3E)-4,8-Dimethyl-3,7-nonadien-1-yl]-5-hydroxy-2,7-dimethyl-2H-chromene-6-carboxylic acid |