Chrysomycin B
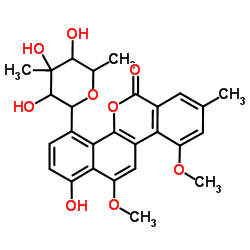
Chrysomycin B structure
|
Common Name | Chrysomycin B | ||
|---|---|---|---|---|
| CAS Number | 83852-56-6 | Molecular Weight | 496.506 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 784.0±60.0 °C at 760 mmHg | |
| Molecular Formula | C27H28O9 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 265.9±26.4 °C | |
Use of Chrysomycin BChrysomycin B is an antibiotic isolated from a strain of Streptomyces. Chrysomycin B causes DNA damage in the human lung adenocarcinoma A549 cell line and inhibits topoisomerase II. Chrysomycin B suppresses the growth of transplantable tumors in mice. |
| Name | Chrysomycin B |
|---|---|
| Synonym | More Synonyms |
| Description | Chrysomycin B is an antibiotic isolated from a strain of Streptomyces. Chrysomycin B causes DNA damage in the human lung adenocarcinoma A549 cell line and inhibits topoisomerase II. Chrysomycin B suppresses the growth of transplantable tumors in mice. |
|---|---|
| Related Catalog |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 784.0±60.0 °C at 760 mmHg |
| Molecular Formula | C27H28O9 |
| Molecular Weight | 496.506 |
| Flash Point | 265.9±26.4 °C |
| Exact Mass | 496.173340 |
| PSA | 138.82000 |
| LogP | 4.99 |
| Appearance of Characters | solid |
| Vapour Pressure | 0.0±2.9 mmHg at 25°C |
| Index of Refraction | 1.663 |
| Storage condition | 2-8°C |
| Water Solubility | DMF: soluble |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
|
Angucyclines: Biosynthesis, mode-of-action, new natural products, and synthesis.
Nat. Prod. Rep. 29(2) , 264-325, (2012) Covering: 1997 to 2010. The angucycline group is the largest group of type II PKS-engineered natural products, rich in biological activities and chemical scaffolds. This stimulated synthetic creativit... |
|
|
A colorimetric microassay for the detection of agents that interact with DNA.
J. Nat. Prod. 55(11) , 1582-7, (1992) A simple microtiter assay for the detection of compounds that bind DNA is described. Agents that displace methyl green from DNA are detected spectrophotometrically by a decrease in absorbance at 630 n... |
|
|
The chemistry of the antibiotics chrysomycin A and B. Antitumor activity of chrysomycin A.
J. Antibiot. 35(9) , 1194-201, (1982) The yellow antibiotic chrysomycin, isolated in crystalline form in 1955, is found to consist of two closely related components, a major one, chrysomycin A, and a minor one, chrysomycin B. They differ ... |
| 1,5-Anhydro-6-deoxy-1-(1-hydroxy-10,12-dimethoxy-8-methyl-6-oxo-6H-dibenzo[c,h]chromen-4-yl)-3-C-methylhexitol |
| Hexitol, 1,5-anhydro-6-deoxy-1-C-(1-hydroxy-10,12-dimethoxy-8-methyl-6-oxo-6H-benzo[d]naphtho[1,2-b]pyran-4-yl)-3-C-methyl- |