(E)-ajoene
Modify Date: 2025-08-25 17:30:49
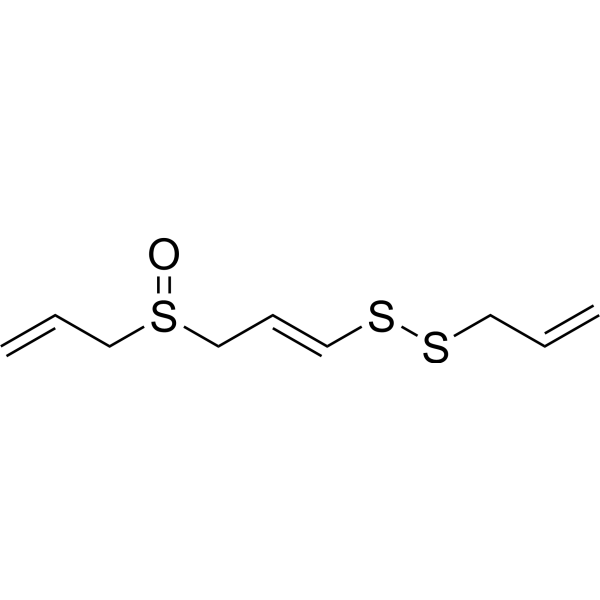
(E)-ajoene structure
|
Common Name | (E)-ajoene | ||
|---|---|---|---|---|
| CAS Number | 92284-99-6 | Molecular Weight | 234.40 | |
| Density | 1.181±0.06 g/cm3 (20 °C, 760 mmHg) | Boiling Point | 376.0±42.0 °C (760 mmHg) | |
| Molecular Formula | C9H14OS3 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of (E)-ajoene(E)-Ajoene is a oxygenated ajoene and organosulfur compound, which can be acquired via allicin decomposing. The polysulfides from garlic can be converted by human red blood cells into hydrogen sulfide (H2S) and allyl glutathione. (E)-Ajoene has been proved to show neuroprotective effects against ischemic damage. (E)-Ajoene is orally active to inhibit lipid peroxidation. (E)-Ajoene increases the number of cresyl violet-positive neurons and decreases the number of reactive gliosis in the CA1 region[1][2]. |
| Name | Disulfide, 2-propen-1-yl (1E)-3-(2-propen-1-ylsulfinyl)-1-propen-1-yl |
|---|---|
| Synonym | More Synonyms |
| Description | (E)-Ajoene is a oxygenated ajoene and organosulfur compound, which can be acquired via allicin decomposing. The polysulfides from garlic can be converted by human red blood cells into hydrogen sulfide (H2S) and allyl glutathione. (E)-Ajoene has been proved to show neuroprotective effects against ischemic damage. (E)-Ajoene is orally active to inhibit lipid peroxidation. (E)-Ajoene increases the number of cresyl violet-positive neurons and decreases the number of reactive gliosis in the CA1 region[1][2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.181±0.06 g/cm3 (20 °C, 760 mmHg) |
|---|---|
| Boiling Point | 376.0±42.0 °C (760 mmHg) |
| Molecular Formula | C9H14OS3 |
| Molecular Weight | 234.40 |
| Exact Mass | 234.02100 |
| PSA | 86.88000 |
| LogP | 3.86790 |
| InChIKey | IXELFRRANAOWSF-FNORWQNLSA-N |
| SMILES | C=CCSSC=CCS(=O)CC=C |
| NSC 614554 |
| (E)-Ajoene |
| Disulfide, 2-propenyl 3-(2-propenylsulfinyl)-1-propenyl, (E)- |
| Disulfide, 2-propenyl (1E)-3-(2-propenylsulfinyl)-1-propenyl |