8-Chloroadenosine-3′,5′-cyclic monophosphorothioate, Rp-isomer
Modify Date: 2023-01-14 09:56:51
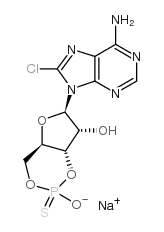
8-Chloroadenosine-3′,5′-cyclic monophosphorothioate, Rp-isomer structure
|
Common Name | 8-Chloroadenosine-3′,5′-cyclic monophosphorothioate, Rp-isomer | ||
|---|---|---|---|---|
| CAS Number | 142754-27-6 | Molecular Weight | 401.69800 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C10H10ClN5NaO5PS | Melting Point | N/A | |
| MSDS | USA | Flash Point | N/A | |
| Name | 8-Chloroadenosine-3′,5′-cyclic monophosphorothioate, Rp-isomer |
|---|---|
| Synonym | More Synonyms |
| Molecular Formula | C10H10ClN5NaO5PS |
|---|---|
| Molecular Weight | 401.69800 |
| Exact Mass | 400.97300 |
| PSA | 182.50000 |
| LogP | 1.62240 |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
|
Modulation of transglutaminase 2 activity in H9c2 cells by PKC and PKA signalling: a role for transglutaminase 2 in cytoprotection.
Br. J. Pharmacol. 171(16) , 3946-60, (2014) Tissue transglutaminase (TG2) has been shown to mediate cell survival in many cell types. In this study, we investigated whether the role of TG2 in cytoprotection was mediated by the activation of PKA... |
|
|
Unhydrolyzable analogues of adenosine 3':5'-monophosphate demonstrating growth inhibition and differentiation in human cancer cells.
Cancer Res. 52 , 2504, (1992) A set of adenosine 3':5'-monophosphate (cAMP) analogues that combine exocyclic sulfur substitutions in the equatorial (Rp) or the axial (Sp) position of the cyclophosphate ring with modifications in t... |
| Rp-8-Cl-cAMPS |
| (4aR,6R,7R,7aS)-6-(6-amino-8-chloropurin-9-yl)-2-hydroxy-2-sulfanylidene-4a,6,7,7a-tetrahydro-4H-furo[3,2-d][1,3,2]dioxaphosphinin-7-ol |