Influence of the solvent in low temperature glycosylations with O-(2,3,5,6-tetra-O-benzyl-β-D-galactofuranosyl) trichloroacetimidate for 1,2-cis α-D-galactofuranosylation.
Gabriel Gola, Mariano J Tilve, Carola Gallo-Rodriguez
文献索引:Carbohydr. Res. 346(12) , 1495-502, (2011)
全文:HTML全文
摘要
Glycosylation studies for the construction of 1,2-cis α-linkages with O-(2,3,5,6-tetra-O-benzyl-β-D-galactofuranosyl) trichloroacetimidate (1) and several acceptors, including D-mannosyl and l-rhamnosyl derivatives were performed. The reactions were conducted at low temperatures using CH(2)Cl(2), Et(2)O, and acetonitrile as solvents. A non-participating solvent such as CH(2)Cl(2) at -78°C, favored the α-D-configuration. In contrast, acetonitrile strongly favored the β-D-configuration, whereas no selectivities were observed with Et(2)O. The use of thiophene as an additive did not enhance the α-D-selectivity as in the pyranose counterpart. Although selectivities strongly depended on the acceptor, trichloroacetimidate 1 constitutes a valuable donor for the synthesis of α-D-Galf-(1→2)-l-Rha and α-D-Galf-(1→6)-D-Man. As these motifs are present in pathogenic microorganisms, these procedures described here are useful for the straightforward synthesis of natural oligosaccharides.Copyright © 2011 Elsevier Ltd. All rights reserved.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
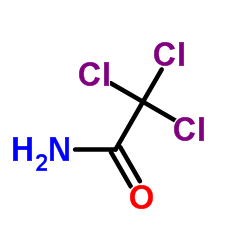 |
2,2,2-三氯乙酰胺
CAS:594-65-0 |
C2H2Cl3NO |
|
An improved synthesis of (-)-5,11-dideoxytetrodotoxin.
2013-02-15 [J. Org. Chem. 78(4) , 1699-705, (2013)] |
|
Activation of glycosyl trichloroacetimidates with perchloric...
2010-09-23 [Carbohydr. Res. 345(14) , 2074-8, (2010)] |
|
Synthetic studies toward the anthrax tetrasaccharide: altern...
2012-07-15 [Carbohydr. Res. 356 , 115-31, (2012)] |
|
Stereoselective rearrangement of trichloroacetimidates: appl...
2014-01-01 [Org. Lett. 11(11) , 2433-6, (2009)] |
|
Rhodium-catalyzed regio- and enantioselective amination of r...
2012-12-07 [Chem. Commun. (Camb.) 48(94) , 11531-3, (2012)] |