Sulphydryl groups in the template-primer-binding domain of murine leukaemia virus reverse transcriptase. Identification and functional analysis of cysteine-90.
S Basu, A Basu, M J Modak
文献索引:Biochem. J. 296 ( Pt 3) , 577-83, (1993)
全文:HTML全文
摘要
Treatment of murine leukaemia virus reverse transcriptase with benzophenone 4-maleimide inactivates DNA polymerase activity, but has no effect on the RNAase H function. Kinetic measurements indicated that benzophenone 4-maleimide is a competitive inhibitor with respect to template-primer binding, but is non-competitive with respect to dNTP binding. Enzyme modified with benzophenone 4-maleimide cannot bind template-primer or primer alone, as judged by u.v.-mediated cross-linking of radiolabelled substrates. Of the eight cysteine residues in murine leukaemia virus reverse transcriptase, only two were modified by benzophenone 4-maleimide, which were identified as Cys-90 and Cys-310 by comparative tryptic-peptide mapping and amino acid composition analysis. Inclusion of template-primer or primer alone in the modification mixture protected only Cys-90 from modification by benzophenone 4-maleimide. To investigate the role of Cys-90 in detail, we converted it to alanine by site-directed mutagenesis. The mutant enzyme, however, exhibited no loss either of DNA polymerase or of RNAase H activity. These results indicate that Cys-90 is located in a domain of murine leukaemia virus reverse transcriptase that binds template-primer, but may not have a direct role in the enzymic function of the enzyme. Ala-90 mutant murine leukaemia virus reverse transcriptase is at least 10-fold more susceptible to heat inactivation than is the wild-type enzyme, which suggests that Cys-90 in murine leukaemia virus reverse transcriptase may play a role in maintaining structural integrity.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
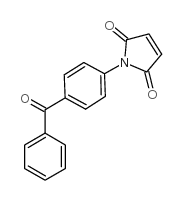 |
4-(N-马来酰亚胺基)二苯甲酮
CAS:92944-71-3 |
C17H11NO3 |
|
Subunit interactions in the clathrin-coated vesicle vacuolar...
1999-10-08 [J. Biol. Chem. 274(41) , 28909-15, (1999)] |
|
Using a low denaturant model to explore the conformational f...
2012-02-21 [Biochemistry 51(7) , 1369-79, (2012)] |
|
A cross-linking study of the N-terminal extension of human c...
2003-09-02 [Biochemistry 42(34) , 10324-32, (2003)] |
|
Formation and properties of smooth muscle myosin 20-kDa ligh...
1991-08-01 [Arch. Biochem. Biophys. 288(2) , 584-90, (1991)] |
|
Identification of the site of photocross-linking formed in t...
1991-02-05 [J. Biol. Chem. 266(4) , 2272-5, (1991)] |